Get Complete Project Material File(s) Now! »
Consequence of stress response to combined stress
Due to the intensity, and duration of stress combination, the consequence of the combined stress varies (Tab. 1). Negative interactions of combined stresses can result in more severe adverse effects on the plant. For example, in Arabidopsis and barley, plants exhibited an aggravating effect on photosynthesis when they were exposed to drought and salinity simultaneously (Ahmed et al., 2013). The adverse effects of nutrient deprivation on plant growth and development are exacerbated when it comes in combination with other abiotic stresses because nutrients are required for the synthesis of ROS (Mittler and Blumwald, 2015; Mittler, 2017). Sometimes the adverse effect is more significant than the addition of two stresses. As a classical case of negative interaction, the antagonistic role in combined stress of drought and heat stress have been studied in many plants (VILE et al., 2011; Prasad et al., 2011; Suzuki et al., 2014; Soolanayakanahally, 2018; Zinta et al., 2018). Under high-temperature stress, plants increase stomatal conductance and thereby reduce leaf temperature through transpiration (Salvucci and Crafts-Brandner, 2004; Way and Oren, 2010). Drought stress usually results in stomatal closure and higher leaf temperature, leading to a decrease in C fixation and thus limiting photosynthesis (Chaves et al., 2003). Taken together, plant growth was significantly reduced under drought and heat stresses and was even more detrimental under their combination (Vile et al., 2012; Prasad et al., 2011).
However, in some cases, combined stress is beneficial to plants in comparison to single stresses. As indicated, heat and drought stress negatively affected Arabidopsis biomass in many ways, but the increase of ambient CO2 concentration significantly alleviated the adverse effects. This mitigation effect of CO2 may be due to the rise of carbon fixation at high CO2 concentrations, which in turn up-regulates the synthesis of antioxidants (polyphenols, ASC and CAT). Through multi-omics analysis, Zinta et al. (2018) revealed that high CO2 alleviates the stress effects by inhibition of photorespiration and H2O2 synthesis. In Medicago truncatula, drought decreases stomatal conductance and promotes the synthesis of ascorbate and glutathione, which in turn improves plant tolerance to ozone (O3) (IYER et al., 2013). Henriet et al. (2020) illustrated that the moderate drought mitigated the negative effect of sulfur deficiency on the accumulation of S-rich globulins in seeds, due to a lower seed sink strength for nitrogen.
Moreover, continuous stress may cause stress memory or priming, which can pave the way to protect plants from future challenges (Hilker et al., 2015; Hilker and Schmülling, 2019). Stress memory is demonstrated to build plant resistance to the very same stress, or even another abiotic stress (Kissoudis et al., 2014; Rejeb et al., 2014). Virlouvet et al. (Virlouvet and Fromm, 2014) revealed that Arabidopsis plants undergoing drought remained at partial stomatal closure during a watered recovery period, facilitating reduced transpiration during subsequent dehydration stress. A systems-biology approach pointed out one gene module that coordinated changes in transcription memory responses established in maize (Virlouvet et al., 2018). It is also shown that pre-harvest UV-C treated plants displayed a stronger resistance to infection in strawberry leaves and led to reduced symptoms (Xu et al., 2019). Likewise, tomato seedlings treated by osmotic stress show adaptation to salinity and drought stress that is mediated by ABA (Andujar et al., 2011). This priming allows plants to respond more quickly to environmental challenges.
It should be noted that plant responses to sequential stresses in any order of the two stresses are often similar. For example, similar adverse effects of physiological limitations were found in the sequential treatment of the two strains in Arabidopsis (Zandalinas et al., 2019), barley (Hordeum vulgare) (Rollins et al., 2013), tobacco (Nicotiana tabacum) and citrus (Citrus reticulata) (Zandalinas et al., 2017), whether drought stress followed heat stress or heat stress followed drought stress. Plants were more severely damaged by combined stresses than by single stress, indicating that the response mechanisms of different plants to combined drought and high-temperature stresses are relatively conserved.
The positive or negative interactions between the combined stresses varied depending on the plant species. Adverse effects were found in wheat when plants were subjected to combined salt and heat stress (Keles and Öncel, 2002). However, the combination of salt and high-temperature stress mitigated the effects of single salt stress on the tomato. Under combined salt and heat stress, tomatoes accumulated large amounts of glycine betaine and trehalose in response, resulting in higher photosynthetic efficiency and leaf water potential. The accumulation of substances such as Glycine Betaine helps to maintain high K+ concentrations, thus ensuring a low Na+/K+ ratio and protecting the plant from salt stress (RIVERO et al., 2013). In addition, Glycinebetaine protects PSII from heat-induced inactivation (Allakhverdiev et al., 2003) and inhibits salt-induced K+ efflux (Cuin et al., 2008; Cuin and Shabala, 2005). Trehalose is important in maintaining photosynthetic capacity (Lunn, 2007) and also contributes to cellular resistance to oxidative stress (Chen and Murata, 2008; Garg et al., 2002). Meanwhile, under combined salt and heat stress, H2O2 accumulation and oxidative protein damage were inhibited, thereby protecting plants from oxidative stress (RIVERO et al., 2013).
In conclusion, plant response to combined stresses needs to be analysed case-by-case. It has been shown that abiotic stress can increase or reduce the resistance of plants to biotic stress. Heat stress leads to the accumulation of reactive oxygen species (ROS) and increases resistance to rice blast in rice (Oryza sativa) (Averyanov et al., 2000); drought and salt stress treatments also increase resistance to pathogens (Achuo et al., 2006). In contrast, abiotic stress has been reported to reduce tolerance to biotic stresses in Arabidopsis. For example, it has been shown that the expression of defence genes is down-regulated in virus-treated plants under combined drought and heat stress, which led to the deactivation of defence responses and higher susceptibility of plants (Prasch and Sonnewald, 2013). Cases of reverse interaction effect existed between N-deficiency and Botrytis infection (Soulie et al., 2020). In addition, high temperature-induced early flowering and suppressed resistance to abiotic stress in Arabidopsis (Liu et al., 2017). Thus, whether abiotic stresses can increase or reduce resistance to another stress depends on the intrinsic, timing, and intensity of the individual stress, as well as their interactions.
Convergent physiological alterations of the combined stress response
When plants are exposed to abiotic stresses, morphological and physiological alteration is mostly expected. Due to the nature of individual stress, how the plants perceive the stress varies. Different abiotic and biotic stress treatment can lead to some common processes as well as unique responses in physiological and molecular aspects.
Specific physiological strategies were commonly employed to respond to stress combinations. Stomatal changes are one of the critical components in regulating this process. The case of heat and drought stress described above has shown a perfect example of the conflicting need for stomatal adjustment toward individual stress. A negative effect on plant photosynthesis is expected when plants limit stomatal conductance in order to reduce transpiration. Another example is when plants were under combined heat and high light stress, both photosynthetic rate and stomatal conductance decreased and leaf temperature increased under high-temperature stress in plants. The effects were more severe under combined drought and high-temperature stress because under drought stress, the stomata of the leaves are usually closed, resulting in lower CO2 availability and limiting photosynthesis (Chaves et al., 2003).
Besides, sugar metabolism is altered in response to environmental stress. Soluble sugars, such as sucrose, fructose and glucose, are not only the metabolic building blocks and energy source but also function as signalling hub that interplay with hormones and nutrients in the fine-tuning of plant growth (Li and Sheen, 2016; Sakr et al., 2018). In cotton (Gossypium hirsutum), soluble sugars could affect auxin biosynthesis through Phytochrome Interacting Factors (PIF) proteins, which lead to anther abortion under heat stress (Min et al., 2014). Additionally, cell wall invertase hydrolyses sucrose and alters carbohydrate partitioning hence modulating defence responses (Tauzin and Giardina, 2014). Downregulation of cell wall-bound invertase expression and activity were observed in combined heat and drought stress (Prasch and Sonnewald, 2013).
Signalling mechanisms of combine stress
To balance the trade-off of fitness costs and stress tolerance, a complex signalling regulatory crosstalk is mostly needed when facing combined environmental stresses. Studies have highlighted several common signalling components including calcium-dependent protein kinase, redox, mitogen-activated protein kinase (MAPK), transcription factors and hormone (Miltter et al., 2016; Zhang et al., 2017).
Second messengers
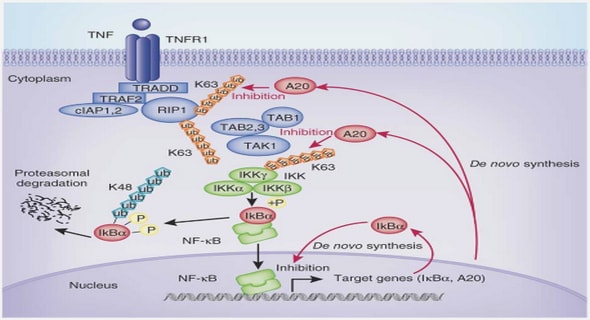
Second messengers, such as Ca2+ signalling pathways (Fig. 1), redox, and electrical signalling networks, play a crucial role in regulating cellular responses to the environment because of the rapid increase after stress stimulation (Gilroy et al., 2014; Ranty et al., 2016). Ca2+ is an essential second messenger for plants and an important regulator of plant growth and development, as well as an essential component of the plant cell wall (Hepler et al., 2005). Ca2+ functions in concert with other critical second messengers like ROS. Osmotic stresses as well as oxidative stresses, heavy metals, and ABA can cause elevated free Ca2+ concentrations in cytoplasm and induce redox perturbations in cells (Zhu et al., 2016). The Ca2+ and ROS signals are usually coupled with the propagation of electrical signals (Choi et al., 2017). A rapid increase in the rate of ROS production occurs as a response to stress conditions. The ROS molecules that mediate signalling functions include hydrogen peroxide (H2O2), singlet oxygen, hydroxyl radical and superoxide anion radical. The activation of mitogen-activated protein kinase (MAPK) cascade by H2O2 and subsequent upregulation of specific stress-related genes in Arabidopsis is a perfect example of ROS-mediated stress-response (Mittler et al., 2004). Nonetheless, Ca2+ and ROS-mediated responses of plants to environmental constraints just form the tip of the iceberg. The mechanism of stress-response in plants is highly intricate and requires several integrated pathways to be activated in response to external stresses. Also, evidence has been found that ROS and NO levels are regulated by auxin. Besides, the GLUTAMATE RECEPTOR-LIKE (GLR) has been reported to play an important regulatory role in electrical and Ca2+-mediated signal transduction (Stephens et al., 2008; Vincill et al., 2012).
Transcriptional regulation and post-translational regulation
Transcription factors (TFs) families (HSF, WRKY, MYB, AP2/ERF, NAC, bZIP, TCP) are common to be induced by several different stress conditions (Zandalinas,2020), such as post transcriptional regulation (Shriram et al., 2016). Integration of various transcriptional networks- that use multiple types of TF family members is needed to mediate combined stress responses. A meta-analysis illustrated that response of different transcription factor families is unique to each stress combination (Zandalinas et al., 2020). For example, HSFs are known to have distinct roles in the regulation of heat stress (Ohama et al., 2017). When heat is combined with other stresses, the upregulation of HSFs is repressed. In addition, for the WRKY gene family, WRKY25, WRKY26 and WRKY33 are positively regulated by heat (Li et al., 2011) while WRKY40 functions antagonistically to AtWRKY18 and WRKY60 to enhance plant tolerance to salt and osmotic stress (Chen et al., 2010). Moreover, ERF family genes were reported to be a positive regulator, not only in salt, drought, and heat stress tolerance, but also in pathogen defence via integrating JA, ET, and ABA signals (Cheng et al., 2013). Illustrated below is an example of an intricate transcriptional network in stress combination (Fig. 2). Additionally, microRNA have emerged as important regulators of developmental and environmental responses at the level of post-transcriptional (Shriram et al., 2016; Xin et al., 2010). For example, miR319 was commonly found to be upregulated under drought, cold, and salt stress (Sunkar and Zhu, 2004). Similarly, a commonly found microRNA, miR164 was reported to be downregulated both in abiotic stresses (salt, mechanical stress) and biotic stress (Tomato Leaf Curl Virus, nematodes) in plants (Hewezi et al., 2008a; Lu et al., 2005; Naqvi et al., 2008).
Transcription factors and molecular responses network during abiotic stress (drought, salinity and cold). Transcription factor-modifying enzymes are shown in squares. Blue squares represent ABA-dependent pathway transcription factors, and orange represent ABA-independent pathway transcription factors. The green boxes represent the cis-elements present in stress-responsive genes. (Modified from (Xie et al., 2019))
Hormone signalling
Interaction between different phytohormone pathways is essential in coordinating tissue outgrowth in response to environmental changes . Among major hormones produced by plants, Auxin is a master regulator that generally influences every aspect of plant growth. Auxin mediates its function mainly due to its interaction with SA and ABA signalling pathways (Park et al., 2009). Additionally, Jasmonic acid (JA) and salicylic acid (SA) are the most important hormones in systemic signalling pathways in response to biotic stress. In addition, brassinosteroid (BR), ethylene (ET) and abscisic acid (ABA) are also involved in regulating systemic signalling pathways (Xia et al., 2011; Gorecka et al., 2014; Gilroy et al., 2016; Proietti et al., 2018; Caarls et al., 2015; Takahashi et al., 2018). ABA is known to stimulate short-term responses like the closure of stomata, resulting in maintenance of water balance. Hormonal signals can also influence the accumulation of microRNA: ABA, cytokinin (CK) and ET negatively impact the accumulation of miR319, while auxin and JA exert a positive impact. Similarly, GA and CK affect miR396 accumulation to activate the ABA pathway (Curaba et al., 2014), which helps to control cell division in response to biotic and abiotic stresses.
An overview of plant hormone signalling networks and their crosstalk in stress responses. ABA, SA, JA and ET are major players in stress response, with ABA mainly regulating osmotic stresses. SA, JA and ET control biotic stress responses. ABA and GA signalling pathways interact, with DELLAs serving as a crosstalk point, to influence the balance between seed dormancy and germination. SA and JA pathways are antagonistically regulated by several transcription factors. JA-ET crosstalk synergistically. Auxins, GAs and CKs participate in biotic stress responses via the SA signalling pathway. CKs also crosstalk with ABA and function in drought and salinity stress responses. Arrows represent positive regulation (accumulation of transcripts, proteins or hormones), and dotted arrows represent negative regulation. (Adapted from (Verma et al., 2016)) Overall morphological and molecular changes are observed in different combined stress, but distinct from the extent of individual stress (Rasmussen et al., 2013; Prasch and Sonnewald, 2015; Olivas et al., 2016). To what extent one stress response will override the other stress response raises the importance of investigation on comparative analysis of individual stress and combined stresses using multivariate approaches.
Nitrogen and water as major limiting factors for plant growth
As the global population continues to increase, biodiversity loss, land degradation and environmental limitations such as water and nutrients have posed a huge threat to crop productivity and food security. In fields, an increasing amount of N fertiliser has been applied to obtain high yields, at the cost of extremely energy-consuming in fertiliser production (Erisman et al., 2008; Chen et al., 2018). Nitrogen and water are major limiting factors for plant growth. Nitrogen is present as the most abundant nutrient in plant tissue among all nutrients plants have to capture from the soil solution while water acts as a solvent, facilitating the mobility of nitrogen and assisting root uptake of nitrogen by shaping the root anatomy.
In nature, crops can be stressed simultaneously by drought and nitrogen deficit, and their interconnection has been emphasised heavily in the field studies (Fig. 4). For rice and other crops, the yield is mainly influenced by water and nitrogen availability. Thus, field management is essential for water and fertiliser inputs. For example, the quantity and timing of irrigation or rainfall need to adjust with respect to N fertiliser application. In non-flooded fields, a sudden rainfall can contribute to decreased N availability by leaching (Fuentes et al., 2008). If fertilised fields experience soil drying, N concentrations in the soil solution will increase, ultimately increasing volatilisation of ammonia from the soil surface. Conversely, if solid N fertiliser pellets are applied to dry soil, less N fertiliser is dissolved. Slow irrigation can reduce volatilisation of N, since N is washed down below the surface, in effect reducing surface N concentrations (Cameron et al., 2013). Indeed, such decisions are essential for more sustainable and better use of N and W application for improving the fate of crop productivity.
Principal processes involved in the response of crop N nutrition to water availability. Dotted arrows indicate fluxes, while plain arrows indicate direct causal relationships. (Modified from Victoria et al., 2010).
Given the critical importance of drought and N-deficiency to plant growth and productivity, it is important to understand the genetic component underlying the stress combined. Large surveys have been demonstrated in the single Nitrogen deficit stress or drought stress episode to understand the stress response strategy in models and crops. The responsive pattern of combined nitrogen deficit and drought stress remains unclear; in the following section, the genomic and functional biological pathways are discussed in the context of drought adaptation, and nitrogen stress will be discussed to shed light on the combinational stress response.
Water and nitrogen stress acclimation
Numerous studies of transcriptome profiling have illustrated this point that when plants are subjected to different abiotic stress conditions, different stress combinations prompt its novel responsive pattern but still keep the single stress response patterns (Prasch and Sonnewald, 2015; Rasmussen et al., 2013).
Physiological and Metabolism coordination
Nitrogen supply can affect stomatal regulation in drought stress
Nitrogen supply can affect physiological status in plants under drought stress. Stomatal pore apertures are generally due to the continuing rise in atmospheric [CO2]. Involvement of environmental signals such as light, [CO2] elevation and the plant hormone ABA can induce rapid stomatal closure. Both soil water deficiency and soil nitrate deficiency can induce stomatal closure and cause reductions in leaf growth rates in plants (Fig. 5A) (Mcdonald and Davies, 1996). When supplied with high nitrate with drying soil, drought stress plants were more sensitive to a decrease in stomatal closure and leaf elongation rates, leading to higher transpiration rates (Fig. 5B) (Wilkinson et al., 2007). Because more Nitrogen supply stimulates more W uptake, and in the meantime, unassimilated N require more CO2 for higher rates of photosynthesis and carbon fixation for assimilation into organic N (Wright et al., 2003; Ding et al., 2018; GUO et al., 2007).
Illustration of nitrogen supply can affect stomatal regulation in drought stress A). Drought stress combined with N-deficiency. B) Ample water with nitrate deficiency C). Drought stress combined with ample nitrate D). The reference condition with ample nitrate and ample water availability.
Table of contents :
Chapter 1 Introduction
1.Plant stress response to combined stress
1.1 Consequence of stress response to combined stress
1.2 Convergent physiological alterations of the combined stress response.
1.3 Signalling mechanisms of combine stress
2. Nitrogen and water as major limiting factors for plant growth
2. Water and nitrogen stress acclimation
2.1 Physiological and Metabolism coordination
2.1.1 Nitrogen supply can affect stomatal regulation in drought stress
2.1.2 Nitrogen regulates root water uptake through aquaporin under drought stress
2.1.3 N metabolism under drought
2.1.4 N assimilation and remobilisation is affected by drought stress
2.2 Signal sensing networks during drought and N deficiency
2.2.1 Specific stress sensor for drought and N deficiency
2.2.2 Common secondary messenger: Ca 2+
2.2.3 Crosstalk of ABA – Nitrate in regulating root growth
3. Systems Biology, an indispensable approach for investigating abiotic stress responses in plants
3.1 multi-omics integration strategy used in plants research
3.2 Multivariate analysis
4. Systems genetics, exploiting natural variation as a source of phenotypic diversity
4.1 Natural variation in stress response: GXE
4.2 GWAS: as a tool to study natural variation
4.2.1 GWAS, a robust mapping method.
4.2.2 Metabolite GWAS use molecular trait as a quantitative trait
4.2.3 Recent approaches to aid GWAS detection.
4.3 Arabidopsis thaliana as a model to study natural variation
4.3.1 From phenotypic variation to genotypic diversity
4.3.2 The complexity of traits’ genetic architecture
4.3.3 Available resource
5 Objective of my thesis
5.1 Identify specific stress response features/pathways in combined stress
5.2 Describe the genetic of their interaction with the environment (G x E)
5.3 Decompose quantitative variation at different levels of complexity (growth, metabolite)
Chapter 2 WxN project
2.1 Phenotypic plasticity among five accessions subjected to N deficiency, mild drought and their combination
2.1.1 N deficiency as well as water deficit affects plant biomass accumulation
2.1.2 Morphological rosette parameters and colour parameters
2.1.3 Physiological parameters are less affected under combined stress than under N deficiency alone.
2.2 Transcriptome changes in plants subjected to N deficiency, mild drought and combined stress
2.2.1 General features in Col-0
2.2.2 Specificities of other accessions
2.2.3 Conserved responses between accessions
2.2.4 General transcriptomic responses to single and combined stress
2.3 Metabolic disruptions caused by stress induced growth limitation among five accessions
2.4 Integrative analyses
2.4.1 Multi-Omics Factor Analysis (MOFA) based on metabolome/transcriptome profiling
2.4.2 Correlation network analysis (WGCNA) failed to identify the most consensual module among accessions
2.4.3 Coexpression networks by Mixomics
2.5 Discussion
Supplementary file
Chapter 3 GWAS project
3.1 Species-wide natural variation in metabolism and growth parameters
3.2 Genome-Wide Association analysis of growth traits and primary metabolism
Test Case: Flowering Time analysis confirms that the DOG1 locus modulates time to flowering
GWAS failed to identify associations with most of the growth traits
GWAS revealed specific metabolic biosynthesis pathways: examples
Colocalized QTL indicates pleiotropic hub controlling central metabolism
GWAS with metabolite ratio identifies genes controlling metabolic architecture
GWAS on principal component traits failed to identify genes comprehensively controlling metabolic architecture and growth
Unknown candidate genes revealed by GWAS
Analysis of KO and HIF Lines for selected candidate genes
3.3 Discussion
Supplementary file
Chapter 4 Methods and material
Plant Materials, Growth Conditions, and Harvest
Relative water content
Total N, Total C
Nitrate analysis
Metabolites Measurements
RNA Extraction and Complementary DNA Synthesis
qPCR
Statistical Analysis
Genome-wide association mapping
Chapter 5 General Conclusion
Reference