Get Complete Project Material File(s) Now! »
CHAPTER 3 RESEARCH METHODOLOGY: QUALITATIVE PHASE
Introduction
This chapter commences with a discussion of the methodology used in this study. First, the main aim and sub-aims as they pertain to the different phases of the research are provided. Next, an overview is given of the four-phase sequential exploratory mixed methods research design that employs a Qualitative phase (Phase 1), a Measuring Instrument Development phase (Phase 2), a Quantitative phase (Phase 3) and a Social Validation phase (Phase 4). The focus of this chapter is on the first two phases, while Chapter 4 is concerned with the Quantitative and Social validation phases. Phase 1 is presented in terms of the aims and steps of the phase, participants, materials and equipment, data collection procedures, trustworthiness considerations, data analysis, and the implications for the next phase. Next, Phase 2, which focuses on the development of the measuring instrument, is presented in terms of the aims and steps of the phase, authentication of the set of hypothetical physical pain scenarios, and material and equipment. Furthermore, the pilot study is discussed in terms of the aims of the pilot study; participants, results and recommendations. Phase 2 concludes with a reflection on the implications for the Quantitative phase.
Aim of the Study
The main aim of this study was to identify the pain-related vocabulary (words and/or phrases) as elicited by specific hypothetical physical pain scenarios in six respondent groups in order to compile and socially validate a list of pain-related vocabulary to be included in an AAC system to assist children who might benefit from AAC to communicate physical pain. The six respondent groups consisted of children aged 6;0 to 7;11; children aged 8;0 to 9;11; parents of children aged 6;0 to 7;11; parents of children aged 8;0 to 9;11; teachers who teach children aged 6;0 to 7;11 and teachers who teach children aged 8;0 to 9;11. The first age group (6;0–7;11) is consistently referred to as the younger group while the second age group (8;0–9;11) is referred to as the older group.
To address the main aim of the study, the following specific aims were formulated for each of the four phases:
(i) Phase 1: To determine and describe those common activities of South African English-speaking children (aged 4;0 to 12;11) that could result in physical pain experiences so as to develop a measuring instrument based on a set of hypothetical physical pain scenarios.
(ii) Phase 2: To develop and pilot test the measuring instrument (HPPS) to elicit pain-related vocabulary, and then to refine the instrument.
(iii) Phase 3: To identify and describe the vocabulary items suggested by the six respondent groups (younger children aged 6;0 to 7;11; older children aged 8;0 to 9;11; parents of younger children aged 6;0 to 7;11; parents of older children aged 8;0 to 9;11; and teachers who teach younger children aged 6;0 to 7;11 and older children aged 8;0 to 9;11) as those words and/phrases that typically developing children aged 6;0 to 9;11 would use to describe physical pain and/or pain-related experiences.
(iv) Phase 4: To determine by means of a stakeholder review of literate adults who use AAC, if the compiled list of pain-related vocabulary (words and/or phrases) would be appropriate for use in scenarios that result in physical pain in order to socially validate the composite list of pain-related vocabulary (words and/or phrases).
Research design
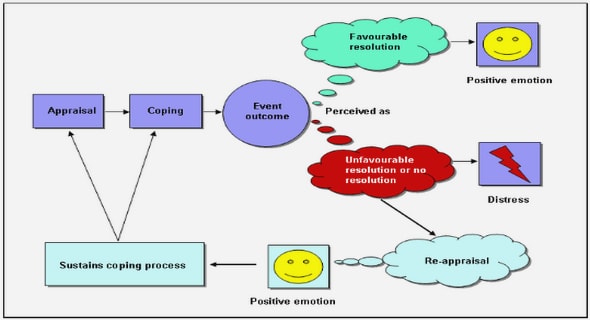
A sequential exploratory mixed method design (Creswell, 2014) was used to address the main aim of the research. The focus of this design is to investigate the phenomenon (children’s pain experiences) qualitatively in an attempt to develop a measuring instrument (a set of hypothetical physical pain scenarios) that can be utilised to gather quantitative data on the given phenomenon (Creswell, 2014). This design was useful in the current study to generalise, evaluate and test the exploratory qualitative results and confirm if they could be generalised to a sample and population (Creswell & Plano Clark, 2011).
The strengths of this design were that during each phase, only one type of data was collected, making it easy to explain, implement, report and produce components of the new measuring instrument as a product of the research process (Creswell & Zhang, 2009). A challenge in the implementation of the exploratory design was that it was time consuming to develop the measuring instrument that involved a set of hypothetical physical pain scenarios (Creswell & Plano Clark, 2011). Previously, two studies used hypothetical pain scenarios with illustrations to evaluate children’s ability to self-report on pain scales, namely the Pediatric Pain Inventory (PPI) (Lollar et al., 1982) and the Charleston Pediatric Pain Pictures (CPPP) (Belter et al., 1988). Due to the fact that both these sets were developed in the United States approximately thirty years ago, and since the method through which the scenarios were developed did not adhere to a rigorous scientific process, it was important to develop an instrument relevant for the contemporary South African context. Therefore, children were involved in sharing their physical pain experiences throughout this study to ensure that a child perspective was obtained (Nilsson et al., 2013). The researcher could afterwards extrapolate qualitative themes and codes from the qualitative phase, which were used as the basis for the development of the quantitative measuring instrument (Creswell & Plano Clark, 2011).
Figure 3.1 illustrates the flow of the sequential exploratory design used in this study. During Phase 1, qualitative data was collected and analysed. Based on this data, the measuring instrument was developed in Phase 2. During Phase 3, quantitative data was collected using the newly developed measuring instrument (Creswell & Plano Clark, 2011) and Phase 4 involved the social validation process. Although this chapter focuses on the qualitative phases (Phases 1 and 2), Figure 3.1 also shows Phase 3 (Quantitative phase) and Phase 4 (Social validation) in order to provide a comprehensive overview
Research Phases
From Figure 3.1 it is clear that the sequential exploratory design as articulated in the present study comprises four phases, namely (a) collection and analysis of the qualitative data (Phase 1); (b) development and pilot testing of the measuring instrument (Phase 2); (c) use of the outcomes of the first two phases to explore the phenomenon quantitatively (Creswell, 2014) and compilation of a list of pain-related vocabulary by means of which children who would benefit from AAC may communicate about their pain (Phase 3); and (d) social validation (by means of a stakeholder review of the proposed vocabulary list) by stakeholders from the specific population for whom it is intended (individuals who use AAC) (Kildea et al., 2011; Schlosser, 1999). The remainder of Chapter 3 will focus on Phases 1 and 2.
Phase 1
Phase 1 is described in terms of the aims; steps; participant selection and descriptive criteria; material and equipment used; data collection procedures; trustworthiness considerations; analysis and results, as well as implications for the next phase.
Aims of Phase 1
The aim of Phase 1 was to determine and describe common activities of South African English-speaking children (aged 4;0 to 12;11) that could result in physical pain experiences so as to develop a measuring instrument based on a set of hypothetical physical pain scenarios.
The following sub-aims were formulated:
(i) To collect drawings made by children of their pain experiences, and to discuss these drawings with the children;
(ii) To identify themes from the children’s drawings and discussions by making a thematic analysis.
Steps of Phase 1
Figure 3.2 outlines the steps that were included in the collection and analysis of the qualitative data of this study. After ethics approval had been obtained from the relevant authorities, parental consent was sought and provided, and children were requested to assent. The children were subsequently requested to draw pictures of activities they had been involved in that resulted in physical pain experiences and they discussed their drawings in focus groups (school setting) or individually (hospital setting). Verbatim transcriptions were made of all the children’s discussions and all transcriptions were checked by an independent person (a qualified teacher with a postgraduate qualification in AAC) who listened to all the audio recordings and compared them for discrepancies with the transcriptions made by the researcher. A thematic analysis was done to compile a list of themes to be included in a set of hypothetical physical pain scenarios.
Participants
Selection Criteria
The criteria for the selection of the two different groups of participants in Phase 1, namely children with typical development in a school setting and paediatric patients in a hospital setting, are set out in Table 3.1. This table also provides a brief description of the methods used to ensure that the criterion was met, as well as a justification for each criterion.
Participant Recruitment
Before recruitment commenced, ethics approval was obtained from the Research Ethics Committee at the Faculty of Humanities, University of Pretoria (Appendix C); the executive head of the independent private school involved in Phase 1 (Appendix D); the chairperson of the Research Operational Committee of the Hospital Company (Appendix E), as well as the hospital manager of the hospital (Appendix E) where data collection took place.
In order to achieve the main aim of Phase 1, two different participant groups who met the criteria outlined in Table 3.1 were recruited to participate. Thirty-eight 6;0 to 9;11 year-old potential participants were recruited from an independent private school in Tshwane South where English was the language of learning and teaching (LoLT) and a Grade R facility was part of the foundation phase. The executive head of the independent private school was contacted telephonically to request permission to conduct research at the school. This request was followed by a formal letter. The executive head, as well as all the teachers of the foundation phase at the school, consented that data could be collected from children in the foundation phase classes of the school. Thereafter the researcher visited the school to distribute consent letters via the children to their parents and inform them about the study (Appendix F). The parents completed the consent form and returned it to the school within a week. Teachers collected the consent forms. A total of 74 consent letters were sent out, 52 were returned (70% return rate), of who 42 gave consent. Four children were absent on the day of data collection, resulting in 38 participants. Before data collection commenced, all the children were asked to assent using a picture-based assent form (Appendix G).
Next, 12 children between the ages 4;0 and 12;11 years who were hospitalised during the time of data collection were recruited from a private hospital in the same geographical area as the school (Tshwane South). These children were recruited because they were able to mention activities related to pain. The hospital manager was first contacted telephonically and then a follow-up letter requesting permission to conduct research at the hospital was sent to her. She provided a letter of consent on condition that the Research Operational Committee (ethics board) of the hospital company would also approve the study. After a formal letter had been submitted to the Research Operational Committee, the chairperson consented that data may be collected at the hospital and stipulated that identifiable information on both the hospital and Hospital Company should be omitted to ensure confidentiality (Appendix E). Consent was granted by the hospital company before data-collection commenced. The parents were with their children at the hospital and were first asked verbally to indicate interest in the study, after which the aim of the research was explained in detail. Once they confirmed, parents received a parental information letter with a consent form (Appendix H) allowing them to consent in writing. All 12 parents consented but insisted that only audio and no video recordings were made, due to the invasiveness of a video recording in a context where the children were vulnerable. All the children assented on the same picture-based assent form used by the participants in the school setting.
Acknowledgments
List of Tables
List of Figures
List of Appendices
Abstract
Opsomming
Chapter 1: Problem Statement and Rationale
1.1 Introduction
1.2 Background and problem statement
1.3 Terminology
1.4 Abbreviations
1.5 List of brand names
1.6 South Africanisms
1.7 Overview of the chapters
1.8 Conclusion
Chapter 2: Literature review
2.1 Introduction
2.2 The history of pain
2.3 Socio-communication model of pain
2.3.1 The internal subjective pain experience of children with significant communication difficulties .
2.3.2 Pain experience and expression
2.3.3 Augmentative and alternative communication (AAC)
2.4 Conclusion
Chapter 3: Research Methodology: Qualitative phase
3.1 Introduction
3.2 Aim of the Study.
3.3 Research Design
3.4 Research Phases
3.5 Phase 1
3.6 Phase2
3.7 Conclusion
Chapter 4: Research Methodology: Quantitative phase
4.1 Introduction
4.2 Phase 3
4.3 Phase 4: Social validation
4.4 Conclusion
Chapter 5: Results
5.1 Introduction
5.2 Data reliability and validity
5.3 Organisation of respondent groups
5.4 Total number of occurrences of pain-related words and/or phrases per respondent group per scenario
5.5 Frequency of the total number of pain-related words and/phrases per respondent group
5.6 Description and refinement of data as pain-related categories
5.7 Statistical inference
5.8 Composite list of pain-related words and/or phrases
5.9 The impact of other factors on children’s use of pain-related vocabulary
5.10 Phase 4: Social validation
5.11 Summary of results
5.12 Conclusion
Chapter 6: Discussion
6.1 Introduction
6.2 Methods for vocabulary selection
6.3 Different perspectives
6.4 Stakeholder review
6.5 Proposed model for selecting vocabulary for sensitive topics
6.6 Conclusion
Chapter 7: Conclusion and recommendations
7.1 Introduction
7.2 Summary of results and the contributions of the study
7.3 Clinical implications of this study
7.4 Critical evaluation of the research
7.5 Recommendations for future research
7.6 Conclusion
References
Appendices
GET THE COMPLETE PROJECT