Get Complete Project Material File(s) Now! »
Chapter 3 Nanoscale distribution of calcium release unit in rats, normal human and diseased human cardiac myocytes
Introduction
The understanding of cardiac disease function acts as a central driver in cardiovascular research. Although animal models have been extensively used to isolate and identify the various causal agents in disease manifestation, it is still however important to appreciate the at times significant differences in normal physiology and time frame of disease exist between animals and humans. To this end, use of human tissues directly provides the best understanding of human disease progression.
Due to the recent development of super-resolution imaging modalities, there has as of yet not been extensive studies into disease formation in human hearts using this technique. The current use of dSTORM and other super resolution setups have already revealed additional detail concerning the nanoscale features of calcium handling structures within rodent models (Y. Hou et al., 2014b; Y. Hou et al., 2015; I. D. Jayasinghe et al., 2012; I. D. Jayasinghe et al., 2014; C. Soeller & Baddeley, 2013; Wong et al., 2013). This is of particular importance as it has been shown that the nanometre detail of calcium regulating proteins is essential for correct cell function (M. B. Cannell et al., 2006).
This chapter therefore aims to use dSTORM techniques as demonstrated previously (Y. Hou et al., 2014b; Y. Hou et al., 2015) on human normal and diseased samples in order to identify possible nanoscale changes to the calcium handling proteins which may impact cell function. Specifically, the RyR protein will be examined in depth and comparisons with previous rodent data will be made to identify possible species variation in CRU organisation.
Use of animal models for disease
Important in evaluating the use of animal models for the understanding of normal and diseased cardiac function within humans is the need for an appreciation of the differences and similarities in physiology between the numerous animal models and humans.
Benefit of animal models
Animal models are currently one of the go to methods for studies into cardiac disease mechanisms. They possess numerous advantages when compared to human subjects. Other than ease of obtaining tissue, the ability to create genetic models and specific disease models allow for the close inspection of disease progression. Within cardiac science, animal models are currently being extensively used in the study of a plethora of different pathologies including; ischemia reperfusion injury (Pedrozo et al., 2010; Zweier, Flaherty, & Weisfeldt, 1987), hypertrophic cardiomyopathy(Geisterfer-Lowrance et al., 1996), and dilated cardiomyopathy(Matsumori & Kawai, 1982). These models have significantly advanced understanding of different aspects of disease formation such as the progressive degeneration of the T-tubular system (Balijepalli et al., 2003; Lyon et al., 2009), and the disruption of calcium release leading to reduction in heart performance (D. M. Bers, 2008; van Oort et al., 2011).
Central to animal models have been the use of murine species. Both rats and mice are the predominant animal models in use today due to their readily available genetic information, short time to maturation, ease in creating genetic mutants, and while showing differences, being relatively similar in physiology due to their mammalian nature (Hasenfuss, 1998). The versatility means that murine models are typically the go-to models for disease progression and experimental drug/therapy intervention approaches (Sakai et al., 1996). The ability to create transgenic strains also makes them particularly useful for single gene knock-in or knock-out experiments typically with the goal to reveal the impact of single proteins on heart function (van Oort et al., 2011). Important of note is the relatively simplistic nature of many of these transgenic lines, where only single genes/protein expressions are typically altered.
The mammalian nature of the murine samples means that the majority of cardiac systems remain essentially the same in humans and murine models. Murine models also possess a similar four chamber arrangement of the heart with distinct separation of the atria and ventricles. Both species additionally show the presence of extensive conduction systems throughout the chambers (Miquerol et al., 2004). At the cellular level key proteins necessary in CICR remain functionally the same with major subcellular structures (such as the T-tubular system) also being present. Localisations of proteins also remain similar with the key CICR associated organelles such as the sarcoplasmic reticulum, and T-tubules localising along the z disc of the sarcomere (Forssmann & Girardier, 1970; Isuru D. Jayasinghe et al., 2014). Intriguingly, the similarities are present to a point where human stem cells can be grafted into rat infarcts to assist healing (Laflamme et al., 2005).
While many similarities exist between murine and human tissue, it is important to note that there are nonetheless many factors which differ between the species. Both physiologically and structurally there are differences which need to be accounted for in models of disease as possible confounding factors. Some of these factors are roughly outlined in the following sections.
Problems associated with animal models
Related to the differences between human and rats are the differences in lifespan and rate of disease progression that occur between species. Current animal disease models often employ genetically knocked in or knocked out models that have rapid disease onset (Hasenfuss, 1998; Pinto, Paul, & Ganten, 1998). While allowing more rapid experiment, this may not accurately mimic human disease which can take many years to fully develop. These models are typically used to model CPVT and associated genetic diseases, however even more heart failure simulating models such as trans-aortic constrictions typically have much faster disease onset than what is expected for humans. The typically late in life onset of human cardiac disease means that there are the additional confounding factors of decades of tissue wear, nutritional differences, and any prolonged medical interventions which are not present in rats.
Cellular variation between human and rat cardiomyocyte
T-tubular difference
The T-tubular system is a complex extension of the myocyte surface membrane which projects to the core of the cell to assist in conducting the action potential deep into the cell. This enables increased synchronisation of excitation contraction coupling (EC coupling) and calcium induced calcium release (CICR.)
There exists a wide variety of T-tubular morphologies. For example, we have shown a highly branched and thin T-tubular arrangement in murine models whereas confocal imaging of humans, pigs and horse myocytes have shown a comparatively simpler network yet much larger individual tubular dimensions (Y. Hou et al., 2015; Isuru D. Jayasinghe et al., 2014). Currently, there is extensive details concerning alteration in T-tubular morphology in in disease progression (Balijepalli et al., 2003; Heinzel et al., 2008; Louch et al., 2006; Lyon et al., 2009), with additional detail regarding variations across different species (I. Jayasinghe et al., 2012; Isuru D. Jayasinghe et al., 2014). However, less detail is known regarding the precise functional effects of altered t-tubule arrangement in between species. In atrial cells, data have suggested an overall reduction in T-tubular network when compared with ventricular cells, with more recent evidence pointing to the presence of moderately extended networks in different species (Smyrnias et al., 2010). In ventricular cells development of T-tubules occurs late in the gestation period, or in the case of rats, in the postnatal period (Brette & Orchard, 2003).
Ryanodine receptor cluster sizes
In terms of raw numbers, Franzini-Armstrong et al provides the closest comparison of release cluster sizes through the use of thin section electron microscopy and the counting of feet observed within dyads (Clara Franzini-Armstrong et al., 1999). What they found when comparing different animals is that, in general, the sizes and density of rat CRU (~188 RyRs) tends to be considerably larger than what is observed in larger mammals such as dogs (~45 RyRs). Unfortunately, this study did not include human samples and relied on estimates on cluster geometry (assumed a circular dyad). Additionally, the small sample size of measurements (n = 30 clusters for rats) leaves considerable uncertainty. More recent estimates of mice couplon sizes (T. Hayashi et al., 2009) suggest ~43 RyR on average per cluster. From light microscopy samples, estimates of RyR cluster size in rats sits typically around ~74 RyR (Chen-Izu et al., 2006) with confocal microscopy and more recently we’ve estimated ~63 RyR using super resolution microscopy (Y. Hou et al., 2015).
Precise cluster measurements of human tissue is still lacking, it is expected that for humans a similar trend should be observed with larger mammals such as dog and rabbit. This is because the T-tubular and calcium release dynamics of the cells much more closely resemble these species (Hasenfuss, 1998; Isuru D. Jayasinghe et al., 2014). Confocal microscopy work on human tissues has shown that this is likely to be the case with greater RyR nearest neighbour separation in human tissue as compared with rat (C. Soeller et al., 2007). This results in an approximate 30% decrease in RyR labelling seen in humans as compared with rats.
Differences in calcium handling
In terms of overall cell calcium transient, there are considerable changes between the rodent species as opposed to human and other larger mammals. Alongside the T-tubular and release cluster changes, the overall calcium flux balance between external and internal calcium stores are different (D. M. Bers, 2000). In rat isolated cell experiments, it has been shown that approximately 8% of cell calcium flux occurs across the sarcolemmal or T-tubular membrane; leaving the remaining 92% of calcium from the internal sarcoplasmic reticulum stores (Bassani et al., 1994). This is in contrast to the larger mammals (in this example rabbit) which show approximately 70% from the SR and 28% from the NCX systems. Humans have similar flux balances to rabbits, though with even more bias towards the NCX pathway of 1:1 extracellular to intracellular (Pieske et al., 1999).
This difference appears to be related to an increased SERCA activity in rodent samples which is matched by a comparative decreased LTCC influx during activation. Influx and efflux must remain balanced to prevent net gain or loss of calcium during the contraction-relaxation cycle and there is evidence showing a greater ICa for rabbit as compared with rats (Yuan, Ginsburg, & Bers, 1996). The origin of this increased SERCA activity is not clear, however, it may be due to an increase in protein levels thereby increasing efflux rate (D. M. Bers, 2000). For rats, the resting SR load is high which occurs due to a slowdown of NCX because of high cellular INa (Shattock & Bers, 1989). When contraction resumes this leads to a larger initial contraction which slowly diminishes forming a negative treppe (staircase) response at low stimulation rates. At faster stimulation rates normal treppe response occurs.
Changes to calcium handling systems in diseased hearts
Dilated cardiomyopathy
Dilated cardiomyopathy (DCM) manifests as a dilation of a pumping ventricle (typically the left) leading to reduced cardiac function. It may lead to subsequent heart failure as the mechanical performance of the heart deteriorates (Roberts, Siegel, & McManus, 1987). The idiopathic form of DCM (IDC) has no clearly discernible cause and presents as a hypertrophy of the heart typically with both ventricular cavities expanded in volume. Progression occurs in a futile cycle where the mechanical stresses upon the heart from the enlarged chamber as a consequence of Laplace’s law leads to further dilation as the heart attempts to compensate (Douglas, Morrow, Ioli, & Reichek, 1989). This dilation further increases wall stress and deterioration. While interventions such as beta blockers have been shown to increase function of the dilated heart (Bristow et al., 1994; Gilbert et al., 1990; Waagstein et al., 1993), it is often not enough to completely halt the progression of disease and heart transplants are often the only viable treatment.
Histological and biochemical studies have shown numerous changes which occur in producing IDC pathology (Brillantes, Allen, Takahashi, Izumo, & Marks, 1992; D. J. Crossman et al., 2011; Lyon et al., 2009). Typically different regions of the chamber will show different extents of functional decline (as measured in terms of shortening.) This feature of IDC has been well documented in many whole heart imaging studies such as echocardiogram (Bach, Beanlands, Schwaiger, & Armstrong, 1995), and tagged MRI imaging (A. A. Young et al., 2001). At the cellular level, changes to both T-tubular arrangement and calcium handling dynamics have been shown to be central in the eventual decline in cellular function (Balijepalli et al., 2003; Ibrahim et al., 2011; Lyon et al., 2009; Wei et al., 2010).
T-tubular changes
Changes to the T-tubular layout have been extensively documented in ICM and other cardiac diseases (Balijepalli et al., 2003; M. B. Cannell et al., 2006; A. Guo et al., 2013; Heinzel et al., 2008; Ibrahim et al., 2011; Lyon et al., 2009; Wei et al., 2010). Disruption of T-tubules has direct follow-on effects on calcium handling by changing the dyadic interface between the T-tubular membrane and the SR membrane where coupling between cell excitation and subsequent contraction occurs (He et al., 2001). Under normal circumstances these membranes are held at nanometre precision to enable the rapid coupling between calcium release from the l-type calcium channel (LTCC) and the calcium induced calcium release from SR via Ryanodine receptor channels (RyR) (M. B. Cannell et al., 2006; D. J. Crossman et al., 2011). The changes in T-tubular membrane structure include an increase in the overall width of the T-tubules, loss of T-tubular features within cells, and loss of T-tubular organisation. These changes may lead to alterations of RyR spatial arrangement and the overall cell calcium transient.
Evidence for the relationship between T-tubular organisation and corresponding shape of the calcium transients from within the cell have been tied in with numerous experiments (Balijepalli et al., 2003; Beuckelmann, Nabauer, & Erdmann, 1992; Brette & Orchard, 2003; M. B. Cannell et al., 2006; Heinzel et al., 2008; Sobie et al., 2006) showing that the close coupling of the T-tubular and SR membrane play an essential role in the control of calcium release within the cell. These changes have been identified as a reduction in peak transient flux, an increased duration of the transient, and an overall reduction in transient decay rate (Heinzel et al., 2008). This is likely to be associated directly with a corresponding decoupling of the RyR release sites from the T-tubular system leading to these receptors being reliant on calcium diffused from coupled sites for release by a mechanism known as fire-diffuse-fire propagation (Dawson et al., 1999) which also underlies the propagation of waves.
A method that has been widely used in the measurement of T-tubular regularity has been the application of Fourier transforms to quantify the loss of axial regularity of tubules (Wei et al., 2010). This method, coupled with direct imaging methods have enabled the characterisation of the extent of T-tubular loss and disorganisation. Within dilated cardiomyopathy, the progressive loss of T-tubules is correlated directly with the corresponding loss of heart function (D. J. Crossman et al., 2011).
The precise cause for the loss of T-tubular system is not well understood, however, Junctophilin (JPH) is implicated as a key protein associated with T-tubular loss. JPH is a membrane protein responsible for the close linking of the SR to the T-tubular membrane by directly bridging across the dyadic cleft (Takeshima et al., 2000). Experiments knocking down the expression of JPH in mice have produced results similar to what occurs during heart failure – an overall reduction in expression of T-tubules and a loss of RyR clustering within the cell (Garbino & Wehrens, 2010). Within human diseased samples, results tend to suggest a decrease in JPH2 protein expression (Landstrom et al., 2007; Minamisawa et al., 2004). There is in addition conjecture regarding the possible cleaving of JPH protein (C. Y. Wu et al., 2014). While this has yet to be tied in with disease models other than acute reperfusion, it may be possible that the cleaving of JPH leads to deregulation of SR – t-tubule linkage further hampering the calcium transient.
Storage and release changes
Occurring in parallel to the changes in T-tubular structure is a corresponding alteration in RyR function and SR calcium load. Hyper-phosphorylation of the RyR protein through chronic beta-adrenergic activation has been documented in heart failure (Ai et al., 2005). The hyper-phosphorylated RyR is shown to be more calcium sensitive (Witcher, Kovacs, Schulman, Cefali, & Jones, 1991) thereby increasing the open probability at rest. This change then results in an increase of diastolic leak from the SR draining the SR and contributing to reduced cellular contractility (Shan et al., 2010).
Structurally, the dyadic clusters formed by RyR on the SR membrane have been shown to change with regards to its association with the T-tubular membrane (Song et al., 2006). One of the central observations is the maintenance of regular RyR arrangement (both longitudinally and transversely) while the T-tubular arrangement becomes disrupted. With the loss of regular T-tubular spacing, the arranged RyR channels become isolated and must function based on a fire-diffuse-fire scenario (Dawson et al., 1999).
Within the SR network, during IDC there are no significant shifts in SERCA expression (Schwinger et al., 1995). However, there is an overall decreased rate of reuptake. The origin of this appears to be in the change of ratio of phosphorylated and unphosphorylated forms of the regulator protein phospholamban leading to greater suppression of SERCA activity (Schwinger et al., 1999). Reduced SERCA2a activity then further exacerbates the already reduced SR load from RyR phosphorylation leading to continued reduction in SR calcium release during systole.
Unanswered questions
While there has been extensive use of rat models in investigating the progression of cardiac disease, less is known about the similarities and differences between subcellular protein arrangements when compared with humans. It has been implicated that subtle changes in cluster arrangement will result in alterations in cell calcium dynamics with smaller dyadic separation allowing better coupling of membrane depolarisation and SR calcium release (M. B. Cannell et al., 2006). As rats and humans possess different dependencies on SR and extracellular calcium sources (D. M. Bers, 2000; Pieske et al., 1999; Shattock & Bers, 1989), as well as extensively different T-tubular arrangements(Isuru D. Jayasinghe et al., 2014), there could likely be differences in nanoscale organisation of RyR into calcium release units.
While extensive research has been documented on the changes in regulation and phosphorylation of the ryanodine receptor protein as a consequence of heart failure (Ai et al., 2005; Sobie et al., 2006; Witcher et al., 1991), structurally, less detail is known. Part of this gap in knowledge originates from the lack of nanoscale structure and which may nevertheless have significant impact on the overall calcium transient (M. B. Cannell et al., 2006). It is difficult to visualise these structures using conventional imaging methods. EM is able to provide high resolution images, however, the ability to label specific protein targets is limited (Clara Franzini-Armstrong et al., 1999). Confocal microscopy has been the main conventional method of imaging proteins (Balijepalli et al., 2003; Chen-Izu et al., 2006; C. Soeller et al., 2007). In the case of cardiac dyads however, it has been shown that the Abbe resolution limit produces images of clusters which lack detail (T. Hayashi et al., 2009).
The advent of super resolution microscopy with resolution beyond the diffraction limit down to single receptor channels provides a means of filling this gap in knowledge. Key factors such as changes in RyR – JPH relationship which have been shown to cause alterations in calcium release (Garbino & Wehrens, 2010; van Oort et al., 2011), and arrangement of the RyR both in terms of distance to adjacent clusters and raw cluster dimensions can also be explored in detail. Use of super-resolution microscopy has already been demonstrated in isolated cells of rat and mice, and was able to reveal the similarities between JPH and RyR distribution, as well as detailed cluster arrangements (Isuru D. Jayasinghe et al., 2014).
Table of Contents
ACKNOWLEDGEMENTS
ABSTRACT
TABLE OF CONTENTS
LIST OF ABBREVIATIONS
CO-AUTHORSHIP FORMS
INTRODUCTION
CALCIUM INDUCED CALCIUM RELEASE AND MYOFIBRILLAR ACTIVATION
MYOCYTE INTERNAL ORGANELLE SYSTEMS
Contractile apparatus
Z disc Signalling
T-tubules
Sarcoplasmic reticulum
GRADED RESPONSE AND TERMINATION OF RELEASE
RYR DISTRIBUTION WITHIN THE MYOCYTE
Peripheral clusters
T-tubule associated release clusters
Non t-tubule associated release clusters .
REGULATION OF THE RYANODINE RECEPTOR PROTEIN
VISUALISATION OF RYR CLUSTERS
Confocal Microscopy
Electron Microscopy
Super-Resolution Microscopy
DSTORM MICROSCOPY
Rationale of dSTORM .
dSTORM imaging
3D dSTORM
dSTORM structural resolution
dSTORM in optically thick sections
SOME OPEN QUESTIONS IN CARDIAC EXCITATION CONTRACTION COUPLING
SCOPE OF THE THESIS
CHAPTER 1
ABSTRACT
INTRODUCTION
MATERIALS AND METHODS
Tissue Preparation.
Immuno Labelling
Super-Resolution Imaging (dSTORM)
Widefield Epi-Fluorescence Imaging
Confocal Imaging
Correlative imaging of diffraction-limited and super-resolution data
Deconvolution
Simulation of dSTORM stochastic effects .
3D rendering
EM imaging
Image analysis for local myofibrillar / inter-myofibrillar space diameter
RESULTS
DISCUSSION
Use of super-resolution for cardiac biophysics studies
Correlative imaging of α-actinin with super-resolution and diffraction-limited confocal microscopies
Resolving sarcomeric structure and non-myofibrillar components
Alpha-actinin, distribution and relationship to sarcomere assembly
CONCLUSION
ACKNOWLEDGEMENTS
SUPPLEMENTARY INFORMATION
CHAPTER 2
ABSTRACT
INTRODUCTION
METHODS
Sample preparation and Immuno-staining
dSTORM imaging
dSTORM Image analysis
Solutions and antibodies
Statistical comparisons
RESULTS
Quantitative super-resolution imaging of RyR distribution
Analysis of RyR superclusters
Relationship of RyR clusters to other myocyte structures
Co-localisation of junctophilin-2 within dyads
DISCUSSION
Super-resolution imaging of internal RyR clusters in transverse sections
RyR cluster shapes and sizes in internal couplings
Small and Large RyR clusters
Super-resolution versus diffraction-limited imaging
RyR clusters and JPH
RyRs and contractile machinery
Limitations
CONCLUSIONS
ACKNOWLEDGEMENTS
TABLES
SUPPLEMENTARY METHODS
Simulated Data
Generation of diffraction-limited images
SUPPLEMENTARY FIGURES
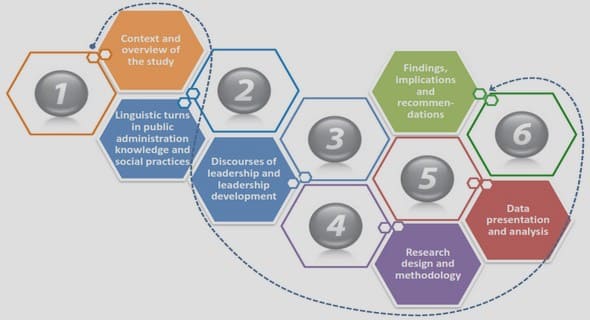
CHAPTER 3 .
INTRODUCTION
Use of animal models for disease
Benefit of animal models
Problems associated with animal models
Cellular variation between human and rat cardiomyocyte
T-tubular difference
Ryanodine receptor cluster sizes
Differences in calcium handling
Changes to calcium handling systems in diseased hearts
Dilated cardiomyopathy
T-tubular changes
Storage and release changes
Unanswered questions
Section aims
METHODS
Tissue preparation and extraction
Immunolabelling
Mounting
Imaging
Image processing
RESULTS
Comparison of Rat and human samples
Transverse imaging
T-tubular morphology
RyR Cluster differences
Colocalisation
Comparison of Human healthy and diseased samples
Cluster morphology changes
Colocalisation changes
T-tubular changes
Numeric Results
DISCUSSION
Comparison of human and rat cardiac myocytes
Calcium release clusters
JPH colocalisation
T-tubular Structure
Human myocytes from normal and diseased hearts
Membrane changes
Changes in RyR labelling density
RyR size distribution
RYR cluster nearest neighbour distance
JPH and RyR relationship
Implications for heart failure
Limitations and Outlook
DISCUSSION
METHODOLOGICAL CONSIDERATIONS
Use of dSTORM for tissue imaging
Comparison of dSTORM with other imaging modalities
Analysis of dSTORM data
IMPACT OF FINDINGS FOR CARDIAC BIOPHYSICS AND PHYSIOLOGY
Alpha actinin and myofibrillar distribution
Effects of RyR Ca2+ release cluster distribution on cell wide calcium dynamics
Relationship between RyR and JPH distribution – impact on cardiac function
Relevance of animal models for release
FUTURE DIRECTIONS
CONCLUSION
LIST OF REFERENCES
GET THE COMPLETE PROJECT
Super Resolution Imaging of Cardiac Ventricular Myocyte Calcium Handling Systems