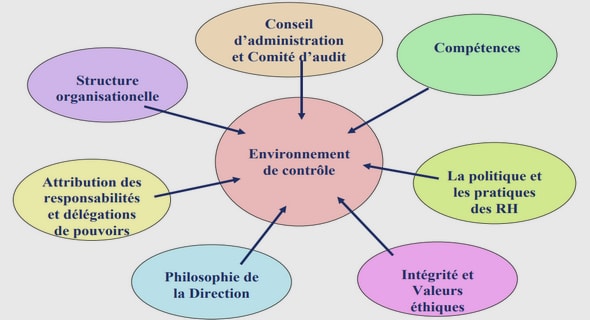
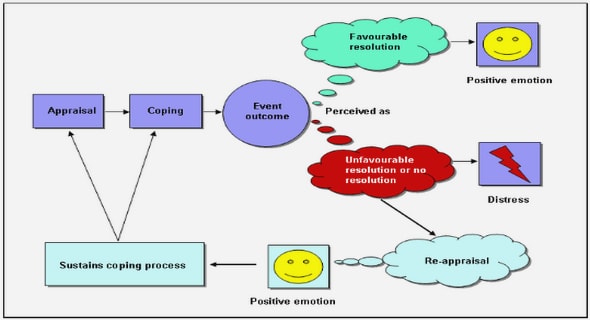
Get Complete Project Material File(s) Now! »
CHAPTER THREE: DENTAL AND SKELETAL INDICATORS OF STRESS
Introduction
Recently the relevance and importance of employing a life course approach in bioarchaeology has been convincingly outlined (Agarwal 2016). In addition, the value that bioarchaeology can bring to life course epidemiology should not be underestimated. Nonetheless, before lifelong associations between early stressors and later adverse health markers in skeletal remains are examined, their benefits and limitations should be carefully considered. This chapter outlines and discusses the nature of various stress skeletal and dental indicators used in this research. The first section deals with indicators reflecting direct physiological health insults associated with infancy and childhood. Often the earliest skeletal indicators are cribra orbitalia and porotic hyperostosis, frequently connected to types of anaemia experienced during subadult development (Stuart-Macadam 1985; Walker et al. 2009; Oxenham and Cavill 2010). Studies that have analysed the timing of active cribra orbitalia and porotic hyperostosis in subadults who lived in industrialised London suggest they most likely reflect stressors operating in infancy and early childhood, specifically between six months and two years (Lewis 1999:177). Therefore, as these lesions might reflect the earliest stressors recorded in this research, they are discussed and analysed first. Enamel hypoplasias are particularly useful as they allow the age of the individual when they experienced stressors to be estimated (Goodman and Rose 1990; Hillson 1996). As hypoplastic defects are the main focus of analysis in this research, studies that have investigated their relationship with survival are discussed at length. Femoral and tibial lengths can provide evidence of chronic growth faltering (Steckel 1995), which may have been influential over a wider age range including adolescence so these are considered last in the series. These early stress indicators are used to assess how developmental environments may have varied for children of differing socioeconomic groups living in London during the Industrial Revolution.
Following on from childhood, direct physiological indicators reflecting adult health risks are discussed. Adult mortality is the definitive stress indicator and is assessed using age at death estimations. A Bayesian approach is used to assign age at death and is discussed further in chapter four. Some forms of skeletal lesions can provide insight into certain chronic conditions and inflammation experienced prior to death. These, in turn, may provide insight into an individual’s general health status. Specifically, an overview is provided for periodontitis, which results from oral bacterial infections (Li et al. 2000), and periosteal new bone formation, which occurs in response to inflammation effecting the periosteum (Weston 2012). Again, these are considered in relation to the frequency and timing of hypoplastic defects to determine if associations might be present, as well as how they might vary by socioeconomic group.
Socioeconomic status is used to assess indirect, or associational, group influences. This operates across the life course, impacting direct physiological factors of both subadults and adults. Information on the socioeconomic positions of the samples used in this research is derived from historical and archaeological sources, discussed in chapter four. Indirect group effects include mortality risks, which are established from mortality profiles. As age at death of the skeletal samples may not accurately reflect the actual risks experienced by the socioeconomic groups, due to the skeletal sample selection criteria, information is obtained for the highest and lowest social groups using burial records for the parish of St. Brides. Collection of these data is also explained in chapter four.
Childhood stress indicators
Cribra orbitalia and porotic hyperostosis
Porotic hyperostosis and cribra orbitalia describe lesions on the cranial vault (frontal, parietals, and occipital) and orbital roofs (cribra orbitalia) that occur as a result of diploic expansion due to marrow hypertrophy (Stuart-Macadam 1985; Ortner 2003; Rivera & Lahr 2017). The lesions, which may be pitted or porous in appearance and may involve raised areas of trabecular outgrowth (Stuart-Macadam 1985), are commonly reported in bioarchaeology investigations and are used to provide insight into diet and disease. Previously, porotic hyperostosis and cribra orbitalia were assumed to both describe the same underlying process of diploic expansion, varying only in the location of the lesions. Recently, however, research using micro computed tomography scans found individuals with cribra orbitalia had significantly thinner diploic bone and thicker outer and inner tables suggesting the two types of lesion may reflect different aetiologies (Rivera & Lahr 2017). Although, in bioarchaeology, both types of lesions are frequently referred to collectively as porotic hyperostosis, here both terms are used to differentiate between them, as their varying distributions are briefly discussed.
In bioarchaeology, active lesions are most frequently reported in crania of infants and children, but rarely in adults, suggesting that in adults they are mostly residual and indicative of anaemia experienced in the first five or so years of life (Stuart-Macadam 1985; Mittler and van Gerven 1994; Lewis 1999; Walker et al. 2009). However, in some cases active lesions are reported in adults, particularly females or individuals of lower status (Sullivan 2005). Hereditary anaemias, such as beta thalassemia or sickle cell anaemia, chronic marrow hyperactivity may also persist into adulthood (Stuart-Macadam 1985; Schultz 2001). In general, cribra orbitalia is more frequently recorded than porotic hyperostosis in adolescence and adults, with active outgrowths of trabecular structures primarily limited to infants and children (Stuart-Macadam 1985; Schultz 2001). The reason why lesions are more common in infants and children is thought to be due to age related changes in marrow composition. From birth, all marrow cavities hold hematopoietic (blood producing) marrow, which is gradually replaced at many sites by non-hematopoietic fatty yellow marrow (Tavassoli 1989). By around four years the replacement of yellow marrow is advanced and by adolescence largely complete, with red marrow sites in adulthood contained to long bone epiphyses, cranium, vertebrae, ribs, and pelvis (Ascenzi 1976). This means that in adults, if increased production of red blood cells (RBC) becomes necessary, yellow marrow can be converted to red (Ascenzi 1976). In young children, however, all sites already contain red marrow, so increased production of erythrocytes is more likely to occur via red marrow hypertrophy. This causes an expansion of diploic bone beyond its normal limits. The result is expansion of the diploe and destruction (osteolysis) of the cortical bone – evident as porosity on the bone surface, with orbital roofs and vault regions particular vulnerable. Evidence from radiographs and skeletal remains suggest that incidences of diploic expansion are most likely to occur between six months and five years of age, with active lesions unlikely after twelve years (Stuart-Macadam 1985; Mittler and van Gerven 1994)
In bioarchaeology, porotic hyperostosis and cribra orbitalia are often considered evidence of iron deficiency anaemia. As iron is a key constituent of haemoglobin (the oxygen carrying protein in RBC) its deficiency is a common cause of anaemia leading to the characteristic diploe expansion (Oxenham and Cavill 2010). However, some researchers have argued that iron deficiency will actually inhibit bone formation and diploe expansion, as iron is necessary for marrow hypertrophy and point to other micro deficiencies as likely causes (Walker et al. 2009). Megaloblastic and haemolytic anaemias are suggested as likely causes of the cranial lesions, as these cause either underproduction, loss, or ineffective RBC, resulting from a combination of poor diet and high disease exposure (Walker et al. 2009), but also some chronic disease conditions (Oxenham and Cavill 2010). With haemolytic anaemia there is abnormal destruction or defective production of RBC, as in thalassemia and sickle cell disease, but this condition can also occur in response to infections, particularly M. pneumonia in children (Elebute and Kesse-Adu 2015). Megaloblastic anaemia is due to impaired DNA synthesis that causes RBC to grow rather than divide and is most commonly attributed to vitamin B12 deficiency (Hoffbrand et al. 2012). Pernicious anaemia, a megaloblastic form, can result from malabsorption of vitamin B12 in the small intestine due to diarrheal disease, parasitic infections, as well as a diets lacking the vitamin (Stabler and Allen 2004). This means groups with low dietary consumption of meat and animal products are particularly at risk, as are infants who are breast fed by mothers with low levels of B12. (Stabler and Allen 2004).
Importantly, recent molecular research has identified a significant relationship between the presence of cribrotic lesions, decreased iron levels, and lower δ15 N isotope values, suggesting the consumption of lower trophic levels foods (Zariſa et al. 2016). While this study confirms a relationship between cribrotic lesions and diet, the authors warn that the decreased iron levels could also reflect vitamin B deficiencies (Zariſa et al. 2016). To further complicate interpretations of cribra orbitalia and porotic hyperostosis, some researchers have pointed out that deficiencies of both iron and vitamin B12 commonly co-occur in individuals (McIlvaine 2015). This could mean that a deficiency in B12, by inhibiting marrow hypertrophy, might mask skeletal evidence of iron deficiencies i.e. diploe expansion. The authors warn that a lack of porotic hyperostosis does not necessarily mean that the individual did not suffer from low iron levels (McIlvaine 2015).
It has been suggested that orbital lesions associated with scurvy (vitamin C deficiency) and rickets (vitamin D deficiency) may be mistaken for cribra orbitalia, as these too can cause porosity in the orbital roofs (Ortner and Mays 1998; Steyn et al. 2016). Although the two conditions frequently co-occur, they can be identified by additional information including the patterning of lesions such as sphenoid porosity in the case of scurvy (Ortner and Ericksen 1997; Brickley and Ives 2006) and flared metaphyses and bone curvature in rickets (Ortner and Mays 1998). As I am analysing adult skeletal remains, it is likely I will mainly encounter residual porotic lesions in the orbits. Without the aid of histological or radiographs, it may not be possible to determine if the residual lesions originated in response to diploic expansion or to chronic bleeds in the superior wall of the orbits, such as with scurvy (Ortner 2003). However, such lesions still provide useful information about the developmental environments experienced by differing socioeconomic groups and point towards variable levels of exposure to disease and poor nutrition during infancy and childhood. So although contention may surround whether porotic lesions are mostly due to iron or vitamin B deficiencies as well as when orbital lesions might reflect other micro nutrient deficits, it is clear that the lesions are all associated with adverse development conditions where both pathogen loads and poor diet are likely to be key. For these reasons, porotic hyperostosis and cribra orbitalia are interpreted more generally in this research as indicators of micro-nutrient deficiencies.
Enamel hypoplasia
Enamel hypoplastic defects are particularly useful nonspecific stress indicators visible in tooth enamel. In the permanent dentition, these developmental defects are records of disturbances in enamel matrix deposition, frequently in response to physiological stressors in infancy and childhood (Goodman and Rose 1990; Hillson 1996). This section will begin with a brief overview of hypoplasia defects before discussing how research has unfolded to substantiate enamel hypoplasia as a reliable gauge of physiological stress. However, research findings that appear contradictory to general trends are also discussed, such as when their occurrences in a population are not associated with decreased age at death. This section concludes with a review of the few studies that have used hypoplastic defects and age at death to explore explanatory models linking early stressors to adverse adult outcomes.
Enamel hypoplasia can present in several forms, including pit, plane or linear types. In bioarchaeology, linear enamel hypoplasia is the most commonly recorded hypoplasia, which appears as horizontal grooves in the enamel surface, usually more pronounced on the buccal or labial surface (Hillson 1996). These result when a band of ameloblasts prematurely cease enamel matrix production before producing the full thickness of enamel (Hillson and Bond 1997; Witzel et al. 2008). Pit type defects are less commonly noted and may present as a single circular cavity, a line of multiple pits, or a scatter of pits over a wider area (Hillson 1996). Plane defects are the least common, appearing as broad or irregular areas of missing enamel often with a marked cervical ledge. Sometimes they reveal an irregular area of exposed striae of Retzius, marked with Tomes process pits (Hillson 1996; Witzel et al. 2008).
Hypoplastic defects can occur in response to systemic insults, trauma to the forming crown, or due to genetic conditions such as amelogenesis imperfecta (Hillson 1996; Brook and Smith 1998). If caused by systemic perturbations, the same insult will register in lateral enamel as a linear groove on all teeth forming at that time, although the prominence may vary depending on the tooth and position thereon (Hillson and Bond 1997). Defects due to trauma can be identified as localised areas of defective enamel, where either the secretory ameloblasts or newly secreted enamel matrix (which is soft and largely unmineralised) has been damaged (Suckling 1980). This can occur when a deciduous tooth is knocked causing its roots to damage the forming permanent crown or if the overlying deciduous tooth becomes infected (known as Turner’s hypoplasia), the inflammation may interrupt normal amelogenesis (Suckling 1980). This type of defect will tend to be localised and only on the affected tooth. It is also less likely to present as a linear defect as direct damage to secretary ameloblasts tends to result in irregular plane type defects, as Suckling (1980) demonstrated using a sheep model. Why some lesions appear as pits is not well understood, but research suggests these might be evidence of more severe stress responses where cohorts of cells have not been able to continue amelogenesis after the stress episode (Witzel et al. 2008). Plane type defects also appear to be caused by severe episodes where a large contingence of cells are permanently interrupted (Witzel et al. 2008). Genetic conditions tend to be rare in most populations and also defects tend to implicate more of the dentition, including discolouration and highly disturbed or missing enamel (Hillson 1996; Brook and Smith 1998; Seow 2014).
In summary, enamel defects that result from systemic stress episodes can be differentiated from those caused by trauma or of genetic origin by their appearance and patterning across the dentition. Linear defects reflect the occurrence of a systemic stress response, evidenced by all ameloblasts at a certain secretory stage being affected and producing a line of defective enamel. The systemic nature of the cellular response is also evident in that all crowns undergoing secretory stage amelogenesis will register disturbed enamel. Tracking these defects across multiple teeth is referred to as chrono-matching. Defect prominence, however, may vary by location within a crown as well as across tooth types – some may only be detectable histologically (Hillson and Bond 1997). Linear defects can be differentiated from ones of traumatic origin as the latter are only on teeth subjected to trauma so cannot be chrono-matched but also tend to present as localised and irregular areas of defective enamel. Genetic defects usually result in obvious generalised enamel deficiencies, such as thin or discoloured enamel with multiple irregular plane type defects affecting most of the dentition (Brook and Smith 1998; Seow 2014).
Enamel hypoplasia was first clearly associated with disease in the 19th century when Hutchinson (1861) suggested their use in diagnosing congenital syphilis. They were later described by Zsigmondy (1893), who first referred to them as enamel hypoplastic defects. Later, experimental animal studies helped to demonstrate a causal relationship between the defects and induced stress, specifically nutritional deficits (Mellanby 1918, 1927; Klein 1931). Schour (1936) first described the neonatal line, a dark band present within the enamel of most deciduous teeth, as being associated with birth. This line was later associated with external hypoplastic defects in children who had suffered birth defects (Kronfeld and Schour 1939). The relationship was further substantiated when histological analysis identified defective enamel, still in the process of forming, along with associated damaged cells in the developing teeth of stillborn neonates (Kreshover 1940). A study by Sarnat and Schour (1941) attempted to directly relate the occurrence and timing of defects with known age of illness, based on medical records, of 60 individuals. Despite assuming that the pace of crown formation is constant and that defect width provides direct information on illness duration, assumptions now known to be incorrect, Sarnat and Schour (1941) were still able to associate just under 50% of the known health insults from systemic and infectious disease with enamel defects. While this study was able to relate many episodes of illness with defect occurrence, it led to the realisation that illness does not always result in a defect and conversely defects did not always have a known cause, at least which could be related to a doctor’s visit. Sarnat and Schour (1941) also limited their study to ten specific systemic and infectious diseases (measles, chickenpox, convulsions, diarrhea, pneumonia, scarlet fever, diphtheria, vomiting, whooping cough, and rickets). This suggested that there may not always be a direct link between specific diseases and the occurrence of hypoplastic defects, pointing to the existence of additional factors influencing their development.
A series of important controlled animal experiments were undertaken in the 1980s that demonstrated how enamel formation was disrupted at a histological level due to trauma and physiological stress (Suckling 1979; Suckling 1980; Suckling et al. 1983; Suckling and Thurley 1984). Of particular interest was one in which lambs were administered various parasite loads to induce a range of systemic stress responses. The lambs were then monitored until death, after which their teeth were analysed (Suckling et al. 1983). This was significant because the research design allowed for causation to be established, as well as an assessment of the dose response between the degree of systemic stress and defect development. The lambs that were administered large doses of a particularly pathogenic parasite experienced severe and acute (7-10 days) diarrhoea and weight loss. All in this group had hypoplastic defects (Suckling et al. 1983: 397). The enamel in lambs that received lower doses of less pathogenic parasites experienced milder, but prolonged symptoms (for example, slight diarrhoea for ~54 days). Defects in these animals did not differ to the controls unexposed to parasites. The study demonstrated that hypoplastic defects visible on the crown surface were only associated with acute and severe systemic disturbances, but not chronic conditions.
Contents
ABSTRACT
ACKNOWLEDGMENTS
CHAPTER ONE: INTRODUCTION
1.1 Introduction
1.2 Aims
CHAPTER TWO: A LIFE COURSE APPROACH TO HEALTH
2.1 Introduction
2.2 A life course approach to adult disease
2.3 Sources of life course data
2.4 Timing and development
2.5 Immunity and weaning
2.6 Critical periods and risk accumulation
2.7 Scarring and selection
2.8 Mortality crossover
2.9 Frailty and the skeletal record
2.10 Conclusion and application to current research
CHAPTER THREE: DENTAL AND SKELETAL INDICATORS OF STRESS
3.1 Introduction
3.2 Childhood stress indicators
3.3 Adult stress indicators
3.4 Conclusion
CHAPTER FOUR: MATERIALS AND METHODS
4.1 Introduction
4.2 Materials and context
4.3 Methods and approaches
CHAPTER FIVE: CHILDHOOD ENVIROMENT RESULTS
5.1 Childhood environments through adult indicators
5.2 Indicators of childhood environment
5.3 The mortality of subadults
5.4 Interpreting variation in childhood environments
5.5 Childhood health experiences in industrialised London
CHAPTER SIX: ADULT ENVIROMENTS
6.1 Introduction
6.2 Periosteal new bone formation
6.3 Periodontitis
6.4 Adult markers of health – an assessment
6.5 Parish burial data
6.6 Measures of adverse health in adult males and females
6.7 Socioeconomic status and adverse health measures
6.8 Adult morbidity and mortality in industrialised London
CHAPTER SEVEN: LIFELONG LINKS BETWEEN THE CHILD AND THE ADULT
7.1 Individual associations from childhood to adulthood
7.2 Childhood health and growth outcomes
7.3 Childhood health and adult morbidity risks
7.4 Childhood health and adult survival
7.5 Lifelong health connections
CHAPTER EIGHT: HOW MIGHT CHILDHOOD STRESSORS INFLUENCE ADULT HEALTH?
8.1 Do childhood stressors impact adult health?
8.2 General impacts of early health insults
8.3 Male and female variation in risks and impacts over the life course
8.4 Influence of Socioeconomic status
8.5 Variation in influences and health outcomes amongst groups.
8.6 The timing of childhood health insults and critical periods
8.7 A stress accumulation model
8.8 Mismatched disease environments
8.9 Extension to risk direction model
8.10 Evaluation of approaches used and further directions
8.11 Conclusion
REFERENCES CITED
GET THE COMPLETE PROJECT
The Cost of Survival: Identifying Life Course Associations between Childhood and Adult Health Outcomes in the Skeletal Record.