Get Complete Project Material File(s) Now! »
Key Biological Concepts
The Brain, Neurons and Synapses
The brain is the center of the nervous system and is the most complex organ in all vertebrates. In all mammals, the outer layer of the brain is denominated as the cerebral cortex. In humans, it can be divided into four distinct lobes: parietal, occipital, frontal and temporal. Each region of the cerebral cortex has its specificities: different cellular composition with different patterns of connectivity.
The cerebral cortex is composed by two main categories of cells, the glia cells (required for structural stabilization and energy supply) and another very heterogeneous class of cells called neurons. Even though recent works have shown the possible implication of glia cells in information processing [1], the main elementary computing units of the nervous system are thought to be the neurons.
Neurons, like all livings cells, present an electric membrane potential, due to the dif-ference between positive and negative charges in the intra- and extra-cellular domain. On the other hand, unlike most cells, neurons are excitable. This means that the electric membrane potential of neurons is capable of fluctuating and, as a consequence, generating and transmitting electrical and chemical stimuli to other cells.
In spite of the large variety of neuronal shapes, a typical neuron can be divided into three structural parts: soma, dendrites and axon. While the dendrites and the axon can be seen as input and output « devices » of the neuron, the soma2 can be seen as the « information processing center » of the cell. This information processing happens in a way that if the total input leads the membrane potential to exceed a certain threshold, then an output signal is generated and transmitted to other neurons by the axon. When this signal is generated, we say that the neuron spiked and that an Action Potential3 was produced. The place where this signal exchange takes place is called synapse. Neurons which are sending and receiving this signal are respectively called presynaptic and postsynaptic neurons.
The central nervous system parts and its units. A) Anatomical parts of the central nervous system. It can be divided into 7 main parts (as indicated in the figure). The brain is the ensemble of three structures: the cerebrum, the cerebellum and the brain stem. The Cerebrum is composed by the cerebral cortex and several subcortical structures (the hippocampus, the basal ganglia, and the olfactory bulb). The Diencephalon is composed by the thalamus, the hypothalamus, the epithalamus and the subthalamus.
Anatomical division of the cerebral cortex into 4 lobes. C) General structure of a neuron. Figures A, B and C were adapted from [2] (pages 9 and 25). or chemical.
In chemical synapses, presynaptic and postsynaptic neurons are completely separated from each other in space. On the other hand, in electrical synapses, pre- and postsynaptic cells are directly connected (sharing their cytoplasm) by means of special channels called gap-junctions.
Gap-junctions have been proposed to contribute to brain rhythms both experimentally and theoretically [3, 4, 5, 6, 7]. However in the network models developed in this thesis, we decided to ignore this level of description, and only included chemical synapses in our models.
Chemical Synapses, Neuro-transmitters and Receptors
At chemical synapses, the synaptic transmission happens by means of vesicles of small organic molecules, called neurotransmitters, released by the presynaptic cell, due to the rise of Ca2+ concentration within the synaptic terminal, following an Action Potential. These vesicles diffuse in the extracellular medium between the pre- and the postsynaptic cells, the synaptic cleft, and bind to the receptors on the postsynaptic neuron. This binding leads to the opening of ion channels at the postsynaptic neuron, generating a change in its membrane potential (see Figure 2). All these processes generate a time difference between the presynaptic spike and the postsynaptic response. This time difference is called synaptic delay.
Synaptic transmission at chemical synapses. A) Membrane potential vari-ation during an Action Potential on the presynaptic cell. B) Succession of events com-posing the chemical synaptic transmission: the action potential arriving at the terminal of a presynaptic axon causes voltage-gated Ca2+ channels at the presynaptic terminal to open (left panel). This opening generates an intracellular high concentration of Ca2+ in the presynaptic terminal, allowing vesicles containing neurotransmitter to fuse with the presynaptic cell membrane and release their contents into the synaptic cleft (middle panel). Subsequently, the released neurotransmitter molecules diffuse across the synaptic cleft and bind specific receptors on the postsynaptic membrane. These receptors cause ion channels to open (or close), thereby changing the membrane conductance and the membrane potential of the postsynaptic cell (right panel). C) Postsynaptic potential fluc-tuation due to the synaptic transmission. This figure was adapted from [2].
Conductance changes by means of chemical synapses can either increase the post-synaptic membrane potential (excitatory synapse) or decrease it (inhibitory synapses), depending on the type of neurotransmitter released. Glutamate and γ-aminobutyric acid (GABA) are respectively the main excitatory and inhibitory neurotransmitters in the brain.
The glutamate-gated channels conduct both N a+ and K+ ions, with nearly equal permeability. The two major glutamate receptors are α-amino-3-hydroxy-5-methyl-4-iso-xazolepropionic (AMPA) and n-methyl-d-aspartate (NMDA) receptors. Unlike AMPA, NMDA channels are additionally highly permeable to Ca2+, which is crucial for induction of synaptic plasticity [8]. While the AMPA-mediated currents are fast (0.34-11ms), the NMDA-mediated are considerably slower (6–350 ms) and present a complex relation with respect to the membrane potential [9, 10, 11, 12]. Both AMPA and NMDA receptors are ionotropic, meaning that these channels open directly when glutamate binds to them. However, the opening of NMDA channels depends not only on the biding of neurotrans-mitters but also on the membrane voltage. At the resting membrane potential extracellular M g2+ ions are tightly bond to NMDA channel pores, blocking ionic current. When the membrane is depolarized (for example, by the opening of AMPA receptor-channels), M g2+ is expelled by electrostatic repulsion, allowing the flux of ions. Due to this M g2+ block, NMDA receptor channels rarely initiate neuronal excitation on their own. The magne-sium block of the NMDA receptor channel (B) is accurately modeled by the following phenomenological expression as a function of voltage of the membrane potential (V) [13] : B(V)= 1 (1) 1 + exp(−0.062V ).([M g2+]o/3.57) where [M g2+]o is the external magnesium concentration (1 to 2 mM in physiolog-ical conditions). NMDA-mediated synapses have been reported to participate in both thalamo-cortical and intracortical synaptic pathways [14] acting on excitatory and in-hibitory neurons [15].
The main receptors of the inhibitory neurotransmitters GABA are GABAA and GABAB. While GABAA receptors are ionotropic, directly opening Cl− channels, GABAB recep-tors are metabotropic, meaning that they activate second-messenger cascades, opening indirectly other channels (often K+ channels). For this reason the current produced by GABAA receptor channels are fast, while the ones from GABAB are slow. Most ofthe fast inhibitory postsynaptic potentials are mediated by GABAA receptors in the central nervous system. In this thesis only GABAA receptors are used in our models. Figure 3 depicts the representative postsynaptic currents due to GABAA, GABAB, AMPA and NMDA receptor channels.
Neuronal Heterogeneity
Although all neurons inherit the same genes, depending on the particular cell’s develop-mental history, only a restricted set of them are expressed. As a consequence, an enormous variety of their enzymes, structural proteins, membrane constituents, ion channels, and secretory products exists among neurons [2, 17, 18, 19]. This rich repertoire of channels allows neurons to generate action potentials with a wide range of shapes (spike waveform), firing rates and duration [20]. These physiological differences can be used to categorized neurons into different types [21, 22, 23, 24]. McCormick and collaborators were the first to describe the so-called Regular-Spiking (RS) and Fast-spiking (FS) neurons. When presented with prolonged stimuli of constant amplitude, RS neurons exhibit pronounced adaptation of the spike frequency while FS neurons undergo little or no adaptation [21] (see Figure 4A and 4B). In 1985, McCormick and colleagues related electrophysiological aspects to morphological and immunocyto-chemical features, identifying neocortical FS neurons as being GABA-mediated interneu-rons [21] and RS neurons as being pyramidal neurons. Today, other types of cell are known to also display regular spiking activity. This is the case for the Cholecystokinin-positive basket cells (GABA-mediated interneuron) [25]. Likewise, two types of GABA-mediated interneurons are known to display fast spiking activity: Chandelier cells4 and Parvalbumin-positive (PV) Basket cells [25]. In this thesis three types of cells are used in our models: Regular Spiking pyramidal cells (RS), Fast Spiking basket cells (FS) and Chattering (Ch) cells.
Chattering Cells were proposed to be a biophysically distinct class of pyramidal neu-rons because of its distinguished intrinsic firing properties [27]. These neurons, found in superficial layers of the cortex, intrinsically generate bursts that repeat with a firing fre-quency of 20 Hz and upwards due to a suprathreshold depolarizing current injection .
Chandelier cells are a type of GABA-mediated interneurons that differs from Basket cells both mor-phologically and functionally. While Basket cells form axosomatic synapses (axons terminating on the cell bodies of target neurons), Chandelier cells form axo-axonic synapses (axons terminating exclusively on the axons of target neurons). Interestingly this morphological difference (inputs onto axon initial segment) proffer this type of cell an excitatory effect on its postsynaptic neurons [26]. Based on the classification by means of the expression of specific molecular markers, Chandelier cells are also classified as Parvalbumin-positive (PV) [25].
A Regular Spiking B Fast Spiking C Chattering
Intrinsic firing patterns of different cell types. A) Extracellular recording in mouse thalamocortical slice of a Regular Spiking (RS) pyramidal cell (adapted from [30, 22]). B) Intracellular recording in guinea pig neocortical slices of Fast Spiking (FS) inhibitory cells (adapted from [21, 22]). C) Intracellular recording in the cat neocortex in vivo of a Chattering (Ch) neuron (adapted from [27]).
Cortical Layers
The neocortex, external region of cerebral cortex5, can be morphologically segmented based on the density of specific pyramidal neurons, as well as by their afferent and efferent projections [31]. Most of the neocortex contains six layers, numbered from the outer surface (pia mater) of the cortex to the inner white matter [2]. Layer L4 receives most of the stimulus coming from the primary sensory thalamic nuclei, together with L5b and L6. Layers L1 and L5a receive inputs from associative thalamus, while layer L5b send projections to subcortical areas. The cortical layers communicate with each other in an intricate way. A scheme of their connections is depicted in Figure 5. In this thesis we are particularly interested in layers 2/3.
Inhibitory neurons are present in all layers and usually have axons that remain within the same area where their cell body are [2]. They are believed to receive inputs from the same sources as the principal cells. It is estimated that inhibitory neurons constitute around 20% to 25% of the neurons in the neocortex.
Brain States : States of Consciousness
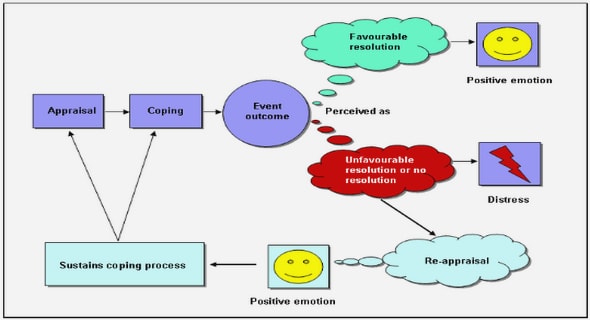
Different brain states can be categorized based on two important concepts: awareness and arousal6 [33]. The level of arousal is associated to the global capacity to respond to stimuli. For instance, clinicians use scoring systems such as the Glasgow Coma Scale [35] to access the level of arousal in patients. In this protocol, the patient reactions (eye opening, motor response and verbal response) are measured with respect to censorial stimuli and scored accordingly. Scores range from 3 to 15, in which 15 indicates a patient fully alert. The level of awareness, on the other hand, is related to the ability to perceive and interact with the environment in a complex and conscious way. In this perspective, awake states are seen as the state in which the patient is fully aroused and fully aware. While a patient in vegetative state is considered to be in a state in which there is some level of arousal, but no level of awareness. The scheme depicted in Figure 6, illustrates these concepts.
In mammals and in most bird species, sleep is divided in two distinguishable neuro-physiological states: rapid eye movement (REM) and non-rapid eye movement (NREM) sleep [36]. REM sleep is characterized by inhibited muscular tone (as measured by elec-tromyography) and involuntary saccadic eye movements (from where its name is derived). In humans, NREM sleep can be divided into three gradual sleep depth levels [37]: stage N1 (transition between wake and sleep), Stage N2 (light sleep) and Stage N3 (deep sleep, also known as Slow Wave Sleep, SWS). The SWS receives this name because of its char-acteristic extracellular activity, which exhibits slow oscillations (as will be discussed in the next section).
In general, the level of arousal increases with the increase of awareness. This is what is observed when we compare the different stages of NREM sleep with conscious wake-fulness. However, an interesting exception exists: REM sleep. This sleep stage presents electrophysiological activity similar to the one observed during Awake states: single units firing tonically, together with extracellular recordings capturing spontaneous bursts of low-amplitude oscillations in the range of 15-90 Hz [38, 39, 40, 41, 42]. For this reason, REM sleep is seen as a state with high awareness but no arousal.
Regardless of the difficulties to rigorously define awareness, some authors tried to quan-tify it, by means of certain concepts of complexity theory. The Complement Research Article inserted in the end of this thesis reviews some of them.
Table of contents :
I Introduction
Key Biological Concepts
The Brain, Neurons and Synapses
Chemical Synapses, Neuro-transmitters and Receptors
Neuronal Heterogeneity
Cortical Layers
Brain States: States of Consciousness
Brain Rhythms
Asynchronous and Irregular States
Gamma Oscillations
Mechanisms of Generation
Gamma Oscillations, NMDA receptors and Schizophrenia
NMDA receptor antagonists in sub-anesthetics doses
Gamma Oscillations in Schizophrenic Patients
Gamma oscillations, psychotic states and NMDAR hypofunction
Computational Models
Historical Perspective
Individual neuron models
Network models and Oscillations
Gamma oscillations in theoretical models
ING Model
PING Model
CHING Model
Human recordings Analysis
Developed Models
II Research Articles
1 Cellular correlates of wakefulness and slow-wave sleep: evidence for a key role of inhibition
2 Integration, coincidence detection and resonance in networks of spiking neurons expressing gamma oscillations and asynchronous states
3 A network model of the modulation of Gamma oscillations by NMDA receptors in cerebral cortex
III Discussion
IV Complement Research Article
Bridging Single Neuron Dynamics to Global Brain States