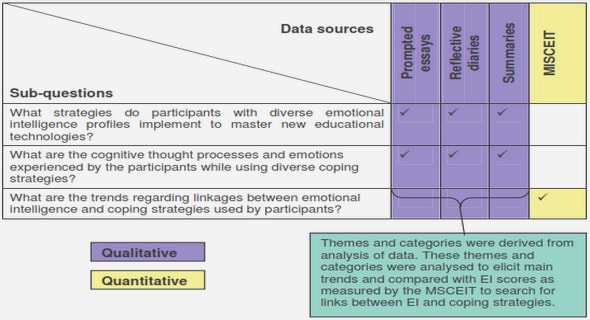
Get Complete Project Material File(s) Now! »
Chapter 3. Identification and Cloning of Alternatively-Spliced Calpain 3 Transcripts.
Calpain 3 Transcripts with Precise Deletions are present in Human Peripheral Blood Mononuclear Cells (PBMCs) but is Absent in Platelets
Calpain 3-specific primers p94-13a and p94-18 flanking the IS2 region designed from the human Calpain 3 sequence (CAPN3 or p94; GenBank Accession number X85030) were used in a PCR based screening of human Peripheral Blood Mononuclear Cells (PBMCs). Products were obtained from human PBMC cDNA that did not correlate with the expected size for Calpain 1 (µ-calpain) and Calpain 2 (m-calpain) of 190 base pairs (bp), or Calpain 3 of 415 bp. Bands of 397 and 283 bp were strongly and reproducibly amplified from PBMC cDNA. Subsequent cloning and sequencing of these products revealed human Calpain 3 specific sequences which possessed deletions mapping to precise exon boundaries within the Calpain 3 transcript. The primers used had successfully annealed to their targets within exons 13 and 18 in the Calpain 3 sequence. The 397 bp fragment showed a deletion of exon 15 ( 15) which is a short 18 bp exon encoding a lysine rich motif considered a potential nuclear localization signal in the mature skeletal muscle Calpain 3 protein. The 283 bp fragment exhibited deletion of both exons 15 and 16 ( 15,16). Both these deletions occurred in the same amplification reaction and consistently mapped to exon boundaries upon sequencing. Both exons 15 and 16 are found within the IS2 insertion sequence of Calpain 3.
PCR amplification of human PBMC cDNA from other, unrelated donors using the same primers revealed an identical pattern of exon deletions of the Calpain 3 transcripts. Subsequent to this, PCR reactions using primers targeted to other regions of the coding sequence of Calpain 3 (primers Cal-3 and p94-7R) revealed the presence of a further deletion within the Calpain 3 transcript, comprising exon 6. This 144-bp exon codes for 48 residues in the IS1 insertion sequence of the mature skeletal muscle Calpain 3 protein. Currently the N-terminal NS sequence and the internal IS1 insertion sequence of the Calpain 3 protein encode motifs with no known protein homologues and are of unknown function. Figure 3.1 compares the results of PCR reactions for the two primer sets that flank exon 6 (Cal-3 and p94-7R) and exons 15,16 (primers p94-13a and p94- respectively, using human PBMC cDNA as a template. Reaction products are also shown for the full length amplicons derived from a mouse skeletal muscle cDNA template. .Since the initial results had been derived from cDNA generated from the total population of PBMCs, an attempt was made to characterize which circulating cell types were responsible for Calpain 3 expression. To answer this question, human PBMC fractions were purified by either Dynabead technology or Fluorescence Activated Cell Sorting (FACS). Whole cellular RNA was extracted from these pools and reverse transcribed to 1st strand cDNA as described in the Materials and Methods section. All cDNAs generated were first tested in separate PCR reactions for the presence of the ubiquitously expressed housekeeping gene, glyceraldehyde-3-phosphate dehydrogenase (mGAPDH). Each cDNA type that produced a characteristic and expected product for mGAPDH indicated successful reverse transcription of cellular RNA and could subsequently be used in PCR reactions targeted to Calpain 3 expression.Fluorescence Activated Cell Sorting (FACS) was utilized to ascertain which PBMC subsets might be contributing to the expression of Calpain 3 transcripts. FACS cells provided were a kind gift from Dr Derek Hart and Dr Georgina Clark of the Haematology Department of the University of the Otago Medical School, Christchurch. The FACS sorted cells were provided as pellets of snap frozen cells on dry ice and included the following cell types. Total ER+ cells 1 x 106 cells total; CD4+, 8 x 105 cells; CD4–, 4 x 105 cells; CD3+, 4 x 105 cells; and CD8+, 1 x 106 cells.In addition Sheep Erythrocyte positive cells (ER+) were provided. These cells consist predominantly of T-cells expressing the CD2+ cell surface marker. These include prothymocytes, immature and mature thymocytes, CD4+ helper/inducer T-lymphocytes and CD8+ cytotoxic T-lymphocytes. Natural killer cells also express CD2+ cell surface markers. ER+ cells showed expression of Calpain 3 transcripts with both exon 15 deleted and exons 15,16 deleted, indicating one or all of the above cell types to be likely candidates for Calpain 3 expression.
Positively selected CD4+ cells (T lymphocyte helper/inducer) were negative for Calpain 3 transcript expression. All other cell types remaining from the CD4+ selection process (the CD4–pool) were tested and produced Calpain 3 transcripts with exon deletion profiles. This result indicates that CD4+ cells comprising Helper/Inducer T-lymphocytes may not express Calpain 3.
PBMC subsets selected on the basis of expression for CD3+ (mature thymocyte) and CD8+ (cytotoxic T-lymphocyte) cell surface markers both proved positive for Calpain 3 transcripts indicating them as a likely Calpain 3 expressing cell types. Figure 3.3 shows a typical result for the FACS sorted cDNAs used as templates in PCR reactions targeted to the exon 15,16 region of Calpain 3 with the p94-13 and p94-18R primer pairs. As can be seen two amplification products are evident in these reactions. The higher molecular weight band of ~400 bp corresponds to the 15 isoform of Calpain 3 while the lower molecular weight band at ~300 bp is that of the 15,16 isoform.
CD19+ Pan-B cells Express Calpain 3.
The PCR-based screening for Calpain 3 transcripts in the FACS sorted PBMC pool involved cell types selected primarily based on T-cell phenotypic markers. A question remained as to whether the B-cell compartment of the PBMC pool may also express Calpain 3. To address this question, pan-B cells were isolated from human whole blood using a Dynabead® isolation protocol 53
specific for the Pan-B CD19+ cell surface marker. Dynabeads® M-450 CD19+ (Pan B) were purchased from Dynal® and used according to the manufacturer’s instructions to separate 10 ml of erythrocyte-depleted human blood cells into CD19+ (Pan B) and CD19– compartments (see Materials and Methods section). Cell counts were taken throughout the extraction procedure with the aid of a haemocytometer. The total number of nucleated cells obtained from 10 ml of whole human blood after erythrocyte lysis, centrifugation at 100 x g and resuspension in 1ml of PBS was 2.5 x 107. Haemocytometer counts of the CD19+ fraction isolated from these cells with the Dynabeads® M-450 indicated the successful isolation of 6 x 105 Pan-B CD19+ cells from the original 10 ml of whole blood. Following this B-cell separation, both CD19+ and CD19– pools were converted to 1st strand cDNA as previously described. In parallel, a complete nucleated blood cell fraction was also harvested for whole cell RNA extraction. Figure 3.4A shows an agarose gel of extracted RNA samples prior to conversion to 1st strand cDNA.
Chapter1. Introduction.
1.1 Background
1.2 Calpain Protein Architecture
1.3 Mechanisms of Action of Calpain 1 and Calpain
1.4 Inhibitors of Calpains
1.5 Metabolic/Physiological Roles and Substrates
1.6 Calpain 3 (CAPN3, p94)
1.7 Biochemistry of Calpain
1.8 Calpain 3 Involvement in Limb Girdle Muscular Dystrophy Type 2A
1.9 Non Skeletal Muscle Calpain 3
1.10 Alternative Splicing and Protein Diversity
1.11 Aims of this thesis
Chapter 2. Materials and Methods.
2.1 Materials
2.2 Methods
Chapter 3. Identification and Cloning of Alternatively-Spliced Calpain 3 Transcripts.
3.1 Calpain 3 Transcripts with Precise Deletions are present in Human Peripheral Blood Mononuclear Cells (PBMCs) but are Absent in Platelets
3.2 Calpain 3 Alternatively-spliced Transcripts are Expressed in T-cell subsets
3.3 CD19+ pan-B cells Express Calpain 3
3.4 Cloning of Full-length Alternatively-Spliced Calpain 3 Transcripts from PBMCs
3.5 Analysis of Murine Tissues for the Expression of Alternatively Spliced Calpain 3 Transcripts
3.6 Cloning of Full Length Calpain 3 from Mouse Adult Skeletal Muscle
3.7 Cloning of an Alternatively-spliced Calpain 3 Transcript from Mouse Thymus
3.8 Submission of Calpain 3 Alternatively Spliced Sequences to GenBank
3.9 Monoclonal Antibodies to Human Calpain 3 Epitopes Detect the Presence of
Calpain 3 Protein Isoforms in Human PBMCs and Blood Cell lines
3.10 Calpain 3 Protein is Detected in Murine Tissues by Western Blotting
Chapter 4. Yeast Two Hybrid Screen for Potential Binding Targets of
PBMC Calpain 3 Alternatively-Spliced Isoforms.
4.1 Introduction to the Yeast Two Hybrid Assay
4.2 Verification of Yeast Host Strains
4.3 Cloning of Calpain 3 Isoforms into pAS2-1-BD
4.4 Transformation of Yeast Host Strains and Confirmation they do not Auto-activate the Reporter Functions
4.5 Bait Construct Toxicity Assessment
4.6 Amplification of the Yeast Two Hybrid Library
4.7 PCR of the Library for the Presence of Calpain 3 Transcripts
4.8 Transformation Efficiency Testing, Cotransformation vs. Sequential
4.9 Calpain 3 Bait Constructs do not Auto-activate the β-galactosidase Reporter Gene
4.10 Yeast Two Hybrid Screen of a Human Leukocyte Library with Human Calpain 3 Δ6,15,16 Using the Host Strain Y190
4.11 β-galactosidase Colony Lift Assay of SD-WLH+ Clones
4.12 Generation of Haploid Y190 Reporter Yeast Containing Library-Derived Target Plasmids by Cycloheximide Curing of the Calpain 3 Bait Plasmid
4.13 Mating Assay to Confirm an Authentic Yeast Two Hybrid Interaction
4.14 Yeast Two Hybrid Screen of a Human leukocyte Library with
Human Calpain 3 Δ6,15,16 Using the Host Strain PJ69-4a 88
4.15 Transformation and Autoactivation Assay of PJ69-4a with Calpain 3 bait plasmids
4.16 Toxicity Assessment for Bait Constructs in PJ69-4a
4.17 Transformation Efficiency Testing of the Library into PJ69-4a
4.18 Screening of the MATCHMAKER™ Human Leukocyte Two Hybrid Library with Human Calpain
4.19 Plasmid Rescue of SD-WLH+ ADE+ Positive Clones
4.20 Mating Assay 94
4.21 Back-Transformation of PJ69-4a Yeast Containing Bait Construct with Library-Derived Plasmids
4.22 GAL4 DNA-BD-Calpain 3 Protein Expression in PJ69-4a and α
Chapter 5. Real Time PCR Quantification of Calpain 3 Splicing Events in
Differentiating C2C12 Myoblasts.
5.1 The Murine Myoblast Cell Line C2C12 Differentiates into Myotubes
5.2 Differentiating C2C12 Cells Display Calpain 3 Alternative-Splicing Events
5.3 Primers Designed to Discriminate Calpain 3 Alternative-Splicing Events in Real-Time PCR
5.4 Control Plasmids for the Testing of Calpain 3 Primers and as Quantification Standards in LightCycler Mediated Real Time PCR
5.5 Quantification of Control Plasmids
5.6 Dilutions Required for Obtaining Plasmid Quantification Standards
5.7 Primer Calibration for Specificity
5.8 Growth and Differentiation of C2C12 Myoblasts and Reverse Transcription to cDNA
5.9 Normalization of C2C12 cDNA with mGAPDH Measurement
5.10 Measurement of Calpain 3 Δ6 Transcripts
5.11 Measurement of Calpain 3 Transcripts Retaining Exon 6
5.12 Measurement of Calpain 3 Δ15,16 Transcripts
5.13 Measurement of Calpain 3 Δ15 Transcripts
5.14 Measurement of Calpain 3 Transcripts Containing Exons 15 and 16
5.15 Normalization of Calpain 3 Transcripts Copy Number to mGAPDH
Chapter 6. Discussion.
6.1 Calpain 3 Transcripts are Alternatively-spliced in Human PBMCs
6.2 Calpain 3 Monoclonal Antibodies Detect Proteins Corresponding to the Δ6,15,16 isoform of Calpain 3 in Human PBMCs and Cell Lines
6.3 Alternatively-spliced Calpain 3 Transcripts in Murine Tissues
6.4 Calpain 3 Monoclonal Antibodies Detect Proteins Corresponding to the Δ6,15,16 Isoform of Calpain 3 in Murine Tissues
6.5 Calpain 3 Isoform Measurements in C2C12 Differentiating Myoblasts
6.6 Homodimerization of Calpain 3 Penta-EF Hand Domain IV
6.7 Homodimerization and Implications for the Failed Yeast Two Hybrid Assay
6.8 Functional Significance of the Unique Calpain 3 Insertions
6.9 LGMD2A and IκBα Mediated Myonuclear Apoptosis
6.10 Future Directions 149
6.11 Implications for Alternative Splicing of Calpain 3 on LGMD2A
Appendices.
GET THE COMPLETE PROJECT
Characterization of Calpain 3 Transcripts in Mammalian Cells: Expression of Alternatively-spliced Variants in Non-muscle Cell Types