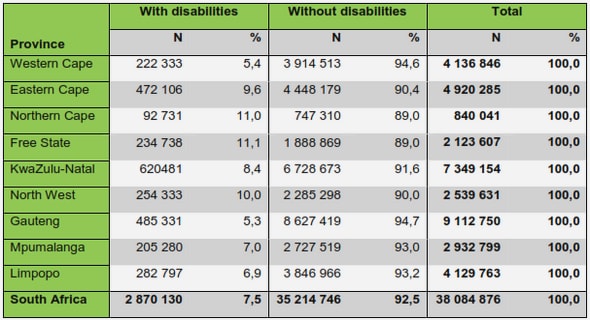
Get Complete Project Material File(s) Now! »
Chapter 3 Electrospun Rubber Fibre Mats with Electrochemically Controllable Pore Sizes
Introduction
Electrospinning is a well-known technique1 that has gained renewed attention with recent innovations in nanotechnology. As discussed in Chapter 1, the process is compatible with large scale production and can create micro- to nano-scale fibres. Electrospun fibrous materials possess several attractive attributes such as a 3D fibrous structure, high porosity and large surface area. While they have been proposed for numerous applications such as desalination membranes2, filters3, 4, polarised light sources5, sensors4, 6, solar cells7 and scaffolds for tissue engineering8, 9, thus far they have mostly been used as passive structural materials since their post-electrospinning structure is fixed. However, there are a few examples were the post-electrospinning morphology is dynamic, such as nanofibre bundle actuators for artificial muscles10 and piezoelectric actuating fibres11.
The post-fabrication control of the structure and other material properties can meet the increasing demand for dynamic micro/nanoscale smart materials, especially in biotechnology, for cell clinics12, pathogen classification13, odour detection14 and within micropumps and micromuscles for microfluidic devices14, 15. It may open new perspectives in numerous applications, i.e. turning (bio)filtration into tunable biofiltration, or mechanically static scaffolds for tissue engineering into electrochemomechanically active stimulating scaffolds. A sensible route to implement such functionality would be the use of CPs, the size and shape of which can be modified under electrical stimulation. Within this class of material, CPs have received increased attention in recent literature16-22, particularly those based on poly(3, 4-ethylenedioxythiophene) (PEDOT)16, 20, 22, 23 and PPy16, 17, 19, 21, 24. When CPs are electrochemically oxidised or reduced, usually at low potentials, insertion/expulsion of counter-ions between an electrolyte and the CP takes place leading to controllable volume changes in the material. These materials combine interesting features, such as softness, lightness and biocompatibility, and show significant promise for applications such as artificial muscles24-31, DNA sensing31-33, and electrochromic devices31, 34, 35. However, CP’s are usually limited by poor mechanical stability, and as such they need to be modified through the use of appropriate dopants36 or through composites34, 37, 38 and additives36. One approach to enhance the properties of CPs is to incorporate them into a cross-linked structure, forming a semi-interpenetrating polymer network (sIPN). A CP sIPN design, directed towards actuator applications, requires specific features. These include having rubbery properties so as not to hinder the electrically driven volume change in the actuating element, and to be cross-linked to avoid flow and creep during actuation. There are several reports on cross-linked electrospun fibres, mainly non-elastomeric fibres that have been cross-linked post-electrospinning, such as in the case of poly(vinyl alcohol)39 (Tg of 85 °C) electrospun fibre mats or biopolymers such as collagen40 or gelatin41. The lack of reports on cross-linked rubber electrospun fibres in the literature is understandable given that rubbers have a glass transition temperature (Tg) below room temperature. This causes the electrospun fibres to coalesce before effective cross-linking takes place and the fibrous morphology is no longer preserved. In the few described cases, butadiene rubber (BR) was cross-linked in situ with UV radiation Here, we report electrospun microfibre mats with dynamic electrochemical stimuli-controllable pore sizes through intrinsic elastomeric and electroactive properties. The material, a sIPN, is comprised of a cross-linked nitrile butadiene rubber (NBR), and an electrically conducting polymer (PEDOT). The elastomeric scaffold is prepared by in situ cross-linking during the electrospinning of the microfibres, and PEDOT is then incorporated via vapour phase swelling of the fibres with the monomer, EDOT, followed by its oxidative polymerisation. The resulting 3D electroactive microfibrous structures are robust, exhibit solvent stability and combine the mechanical properties of the electrospun fibres and the electromechanical properties of the conducting polymer. The change in the pore sizes was characterised though confocal microscopy with in situ electrochemical stimulation in an organic electrolyte as well as in biologically important phosphate buffered saline (PBS) solution.
Materials and Methods
Chemicals
NBR with 44% acetonitrile (ACN) was kindly provided by Lanxess (Mw = 230 000 g mol-1, Per-bunan 4456F). Chloroform, methanol, benzoyl peroxide (BPO), poly(ethylene glycol) dimethacrylate (PEGDM, Mn = 550 g mol-1), poly(ethylene oxide) (PEO, Mn = 100 000), 2,2-dimethoxy-2-phenylacetophenone (Irgacure 651), propylene carbonate, copper(II) chloride, tert-butyl acrylate (tBA), N,N,N′,N′′,N′′-pentamethyldiethylenetriamine(PMDETA), tin(II) ethylhexanoate, acetone, anisole, hydroxyl methyl 3,4-ethylenedioxythiophene, 2-bromopropionyl bromide, 4-(dimethylamino)pyridine, lithium perchlorate (LiClO4), acetonitrile, methane sulphonic acid, propylene carbonate and phosphate buffered saline (PBS) solution were purchased from Sigma Aldrich. Anhydrous iron III chloride was purchased from Acros.
3,4-Ethylenedioxythiophene (EDOT) (H C Starck) was distilled under reduced pressure before use. Lithium bis-trifluoromethanesulfonimide (LiTFSI) was purchased from Solvay. All materials were used as received unless otherwise stated.
Electrospinning NBR/PEGDM
The electrospinning solutions (Table 3. 3) were prepared by dissolving NBR in chloroform or THF until homogenous solutions were obtained. This is followed by the addition of the corresponding amounts of poly(ethylene glycol) dimethacrylate PEGDM (which was run through a basic activated alumina column before use to remove the inhibitor MEHQ), poly(ethylene oxide) (PEO), Irgacure 651 and benzoyl peroxide (BPO). The solution was further stirred until it became homogeneous and drawn into a 1 ml or 5 ml all glass syringe with an 18G metallic needle. The electrospinning was then performed using 8 to 14 kV electric field, with a 10 cm needle-collector distance and 1.0 ml/h pump rate. In situ cross-linking was achieved with the photoinitiator, Irgacure 651, and using an 80000 µW/cm2 UV lamp purchased from South West Pacific (model SB100P/FA). Further cross-linking was achieved though thermal initiator, BPO, in a vacuum oven at 80° C under reduced pressure.
Synthesis and characterisation of PEDOT embedded microfibres
PEDOT incorporation
The electrospun mats were swollen with 3, 4-ethylenedioxythiophene (EDOT) using vapour phase deposition for 2-6 hours achieving a swelling ratio of 80 – 120%. EDOT was then oxidised via immersing the swollen electrospun mat in an aqueous solution of 1.5 M FeCl3 for various times at 40 ° C (Figure 3.6), resulting in polymerisation of the EDOT and forming the PEDOT interpenetrated elastomeric microfibres.
SEM and EDX analysis
SEM on samples with and without PEDOT was conducted with a Philips XL30S FEG with a SiLi (Lithium drifted) with a Super Ultra-Thin Window EDX detector and with a LEICA LEO S430i SEM. The samples prior to PEDOT incorporation were coated with a Quorum Q150RS sputter coater to give a double layer of platinum coating. Samples for EDX and cross-section SEM images were cryocut in liquid nitrogen before imaging.
Elemental analysis of PEDOT embedded microfibre mats
Elemental analysis was carried out by the microanalysis department of ICSN-CNRS. The tests were made 3 times per sample in order to obtain an average.
Electrochemical characterisation of the PEDOT embedded microfibre mats
Cyclic Voltammetry was carried out using a VSP potentiometer (Biologic) at 50 mV/s for samples in 1 M LiTFSi PC solution and PBS solution. For the samples in PBS solution the rate was also recorded at 5, 10, 20, and 100 mV/s. The fibre mats were placed in a gold mesh that was then used as the working electrode. Platinized titanium wire and silver wire were used as counter and reference electrodes, respectively. Conductance measurements were carried out using a Keithley 2000 multimeter with gold contacts.
Confocal microscopy analysis of PEDOT embedded microfibre mats
Pore size variation was visualised in situ by immersing the film in 1 M LiTFSI – PC solution or a PBS solution and using a confocal microscope to map and monitor the changes. The confocal microscope used was an inversed ZEISS Axio Lab A1 microscope with an x63 oil Plan Apo lens. The sample was in contact with a gold mesh as the working electrode under the control of an Autolab PGSTAT101 potentiostat, and used in con-junction with a platinized titanium counter electrode and a silver reference electrode. The gold mesh was separated from the counter electrode by a poly(vinylidene fluoride) PVDF membrane. During the experiment potential was stepped from -0.5 to +0.6 V. Image analysis for SEM images and confocal microscopy images was performed using the ImageJ software package.
Bulk IPN synthesis and characterisation
Non-electrospun bulk film samples were prepared from a solution containing 40 %wt polymer and 60 %wt CHCl3. The polymer weight percentage was comprised of NBR and PEGDM at a 100:56 ratio respectively. The photoinitiator (Irgacure 651) was then added at 5%, 10% and 20% of the NBR mass for the different samples. The samples were prepared by setting the resulting viscous solution between two glass slides, separated by a 250 µm Teflon seal, clamped together and passed under a high intensity UV light source (100 Wcm-2) for 200 sec or for 300 sec. Soxhlet extraction was then performed in chloroform for 3 days in order to obtain the extractable content. The remaining samples were used for characterisation by dynamic mechanical analysis (DMA) and ionic conductivities in 1 M LiClO4 PC solution, PBS solution, and in 1 M LiTFSI PC solution. Ionic conductivity of the non-electrospun samples was performed using a VSP potentiometer (Biologic) over a frequency range from 0.01 Hz to 1 MHz. A perturbation of 10 mV was applied to the cell consisting of the sample sandwiched between two stainless steel electrodes. The conductivity value was calculated from the bulk resistance in the complex impedance diagram. During the test, the temperature was controlled by means of programmable hot plate cells. The samples were swollen in the corresponding electrolyte solution overnight before performing impedance measurements. The values were recorded 3 times and averaged to take into account solvent evaporation.
Results and Discussion
Electrospinning of elastomeric microfibres
While literature on most electrospun rubbers use THF as a solvent42, 43, this was found to be a poor solvent for the system reported here (Figure 3. 1 A, C and E). Chloroform was found to be far better suited. Thus, the electrospun elastomeric scaffold was fabricated from an optimized solution of CHCl3 containing linear high molecular weight NBR and poly(ethylene glycol) dimethacrylate (PEGDM). NBR provides the rubbery properties while the PEGDM acts as a cross-linker. PEGDM was originally intended to have a much higher weight ratio, however due to its poor performance in electrospinning this was reduced. PEGDM was expected to facilitate the subsequent PEDOT interpenetration into the NBR network due to the compatiblising effect of ethylene oxide groups present on both the PEO and the PEDOT repetitive units45, 46. Also, if the PEGDM content was higher it is expected that this would improve hydrophilicity and ionic mobility.47 These materials were chosen due to their success in literature, specifically in the use of forming PEDOT based sIPN actuators.47, 48
The glass transition temperature of NBR and PEO is below room temperature (Tg = -10 °C and -40 °C respectively) and PEGDM is in a liquid state at the room temperature. Therefore, in order to prevent coalescence of the electrospun fibres, in situ cross-linking had to be performed during electrospinning. The free radical, UV photoinitiator (Irgacure 651) was added into the electrospinning solutions to cross-link the polymers during the electrospinning process49. The solution viscosity is a major variable in determining the fibre morphology during electrospinning and contributes to the fibre thickness and fibre beading.50, 51 The PEGDM used in this work had a Mn of 550 g mol-1. It is a liquid with low viscosity (6.5 mPas-1) at room temperature and thus did not contribute to the viscosity as much as the high molecular weight NBR. The PEGDM concentration was explored first, however the resulting solutions with higher PEGDM content quickly showed poor electrospinning qualities (Figure 3. 1 J and L) and thus the concentration was fixed at 56 %wt vs NBR. While the fibres including PEO showed favourable morphology (thin, uniform non-fused fibres), PEO did not crosslink and thus the fibres were not stable to the temperature and solvents necessary to embed PEDOT. Addition of PEO and the optimum ratio in regards to stability was investigated further in fabrication of aligned electrospun fibres (see Chapter 4).
The NBR concentration in the electrospinning solution was then explored, while keeping the PEDGM proportion at 56 %wt/wt vs NBR (Table 3. 3). The PEGDM ratio was set at this value in order to increase and optimize the compatibility with the PEDOT facilitated by the ethylene oxide group. Large beading occurred at 5% wt/vol of NBR (Figure 3. 1 D), whereas much more uniform fibres were seen at 10% wt/vol with fibre diameters ranging from 3 to 6 µm. It was therefore decided to continue further experiments with 10% wt/vol of NBR.
The influence of the cross-linking initiators (Irgacure 651 and BPO) concentration on the fibre morphology was then studied. While low concentrations of the UV initiator (5 and 10 %wt vs NBR) produced decent fibres (creating a porous mat useful for applications such as filtration) (Figure 3. 1 H and I respectively), the mats produced from these compositions lost their fibrous morphology upon solvent-facilitated detachment of the mats from the collector, indicating that they were insufficiently cross-linked. At higher initiator concentrations (20 %wt photoinitiator vs. NBR) the resulting fibre morphology was inconsistent, with sporadic domains of highly fused fibres and relatively non-fused fibres (Figure 3. 1 L and M). This was attributed to cross-linking occurring inside the electrospinning syringe, which varied the solution composition that formed the electrospinning jet with time (i.e. a decreasing polymer concentration with time due to cross-linked polymer becoming trapped within the syringe). A free radical initiator for thermally induced polymerisation, benzoyl peroxide (BPO), was therefore added to the electrospinning solution in order to induce further thermal cross-linking in addition to UV-initiated cross-linking. The thermal cross-linking of the UV-cross-linked electrospun mats was performed at 80 °C in a vacuum oven after electrospinning. While no obvious improvement in morphology was observed with the addition of the thermal initiator when a low UV initiator concentration (5 %wt Irgacure 651, and 5 %wt BPO) was employed (Figure 3. 1 N), quite uniform fibres (Figure 3. 1 O) were produced with a moderate UV initiator concentration (10 %wt Irgacure 651 and 5 %wt BPO). Increasing the concentration of both initiators further (20 %wt. Irgacure 651, 10 %wt. BPO) produced well-defined fibres, as depicted in Figure 3.1 K. However significant cross-linking occurred in the syringe, which caused the syringe to jam, this led to difficulty in producing free-standing fibre mats.
Table of Contents
Abstract
Publications
Acknowledgements
Table of Contents
List of Figures
List of Abbreviations
Co-authorship Forms .
Chapter 1: Introduction
1.1 Brief Introduction to Conducting Polymers
1.2 Electrospinning
1.3 Tissue engineering
1.4 Macromolecular Engineering
1.5 Sensors, Conducting Polymers and DNA sensing
1.6 Objectives
Chapter 2: Applications of Electrospun Conducting Polymer Materials
2.1 Electrospinning and Conducting polymers
Chapter 3: Electrospun Rubber Fibre Mats with Electrochemically Controllable Pore Sizes
3.1 Introduction
3.2 Materials and Methods
3.3 Results and Discussion
3.4 Conclusions
Chapter 4: Electrospun Conductive Cardiac Patch
4.1. Introduction
4.2 Experimental Section
4.3. Results and Discussion
4.4 Conclusion
Chapter 5: Enhanced DNA Sensor with PEDOT Embedded Electrospun Fibres
5.1 Introduction
5.2 Materials and Methods
5.3 Results and Discussion
5.4 Conclusion
Chapter 6: Gold Nanoparticle Decorated Electrospun Fibres
6.1 Introduction
6.2 Experimental
6.4 Conclusions
Chapter 7: Conclusions and Outlook
7.1 Summary of Scientific Contributions of this Thesis
7.2 Outlook and Future Work
References .
GET THE COMPLETE PROJECT
Electrospun Conducting Polymer Rubber Fibres: Synthesis and Applications