Get Complete Project Material File(s) Now! »
CHAPTER 2 Introduction
Electrochemistry: An Overview
Basic concepts
Electrochemistry, as the name suggests, studies the relationship between electrical and chemical occurrences.1 It covers mainly two areas: electrolysis-conversion of chemical compounds by passage of an electric current and electrochemical power sources-energy of chemical reactions transformed into electricity. Electrochemistry is one effective technique to study electron transfer properties. When electron transfer is between a solid substrate and a solution species, it is termed heterogeneous process. Inversely, if electron transfer reaction occurs between two species, both of which are in solution, the reaction is homogeneous. Therefore, electrochemistry can specifically be defined as the science of structures and processes at and through the interface between an electronic (electrode) and an ionic conductor (electrolyte) or between two ionic conductors.2
Electrochemistry is an interdisciplinary science that is mainly rooted in chemistry and physics; however, also linked to engineering and biochemistry/biology. Due to its diversity, electrochemistry has found application in various working fields such as: analytical electrochemistry; environmental electrochemistry;3 electrochemistry of glasses;4 ionic liquids electrochemistry; microelectrochemistry,nanoelectrochemistry; photoelectrochemistry;3 solid state electrochemistry; solution electrochemistry; surface electrochemistry; technical electrochemistry with numerous subtopics such as: Batteries and energy storage5; Corrosion and corrosion inhibition; Electrochemical synthesis and reactors (electrosynthesis); Electronic components (capacitors, displays, information storage, rectifiers); Fuel cells and galvanic deposition; Theoretical or quantum electrochemistry. Electrochemistry also involves the measurement of potential (potentiometry) or current response (voltammetry).6 The work described in this dissertation involves current measurement, voltammetry and a number of voltammetric techniques, namely, cyclic voltammetry (CV), linear sweep voltammetry (LSV), square wave voltammetry (SWV), chronoamperometry (CA) and chronocoulometry.
The electrode-solution interface
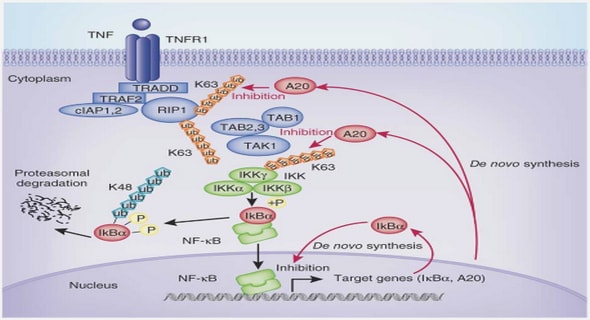
The interface between a metal (electrode) and an electrolyte solution is considered when trying to gain an impression of the structures and processes that occur in electrochemical systems. Figure 2.1 shows a schematic diagram of the interface structure and the processes. This interface is generally charged; the metal surface carries an excess charge, which is balanced by a charge of equal magnitude and opposite sign on the solution side of the interface. The resulting charge distribution; two narrow regions of equal and oppositecharge, is known as the electric double layer. It can be viewed as a capacitor with an extremely small effective plate separation, and therefore has a very high capacitance.
Reactions involving charge transfer through the interface, and hence the flow of a current, are called electrochemical reactions. Two types of such reactions can occur at the interface. One is an instance of metal deposition. It involves the transfer of a metal ion from the solution onto the metal surface, where it is discharged by taking up electrons. Metal deposition takes place at specific sites. The deposited metal ion may belong to the same species as those on the metal electrode, as in the deposition of a Ag+ion on a silver electrode, or it can be different as in the deposition of a Ag+ion on platinum. In any case the reaction is formally written as:
Ag+(solution) + e- (metal) Ag(metal) (2.1)
Metal deposition is an example of a more general class of electrochemical reactions, ion transfer reactions. In these an ion, e.g. a proton or a halide ion, is transferred from the solution to the electrode surface, where it is subsequently discharged.
Another type of electrochemical reaction, is an electron-transfer reaction, an oxidized species is reduced by taking up an electron from the metal. Since electrons are very light particles, they can tunnel over a distance of 10 Å or more, and the reacting species need not be in contact with the metal surface. The oxidized and the reduced forms of the reactants can be either ions or uncharged species. A typical example for an electron-transfer reaction is:
Fe3+(solution) + e- Fe (metal) Fe2+(metal) 3+(solution) + e- (metal) Fe2+(metal) (2.2) Both ion and electron transfer reactions entail the transfer of charge through the interface, which can be measured as the electric current. Based on the definition of the electric double layer, Helmholtz7 proposed a simple geometric model of the interface. This model gave rise to the concept of the inner and outer Helmholtz planes (layers). The inner Helmholtz layer comprises all species that are specifically adsorbed on the electrode surface. If only one type of molecule or ion is adsorbed, and they all sit in equivalent positions, then their centres define the inner Helmholtz plane (IHP). The outer Helmholtz layer comprises the ions that are closest to the electrode surface, but are not specifically adsorbed. They have kept their solvation spheres intact,and are bound only by electrostatic forces. If all these ions are equivalent, their centres define the outer Helmholtz plane (OHP). The layer which extends from the OHP into the bulk solution is a three dimensional region of scattered ions called the diffuse or Gouy layer. The IHP and OHP represent the layer of charges which is strongly held by the electrode and can survive even when the electrode is pulled out of the solution.
DEDICATION
DECLARATION
ACKNOWLEDGEMENTS
ABSTRACT
Table of Contents
Abbreviations
List of Symbols
List of Schemes
List of Tables
List of Figures
CHAPTER 1 General Overview of Dissertation
1.1 Background of Project
1.1.1 Introduction
1.1.2 Aim
1.1.3 Objectives
1.1.4 Scientific novelty of the work
1.1.5 Publication of the work
1.1.6 Structure of the thesis
References
SECTION A
CHAPTER 2
Introduction
2.1 Electrochemistry: An Overview
2.1.1. Basic concepts
2.1.1.1. The electrode-solution interface
2.1.1.2. Faradaic and non-Faradaic processes
2.1.1.3. Mass transport processes
2.1.2. Voltammetry
2.1.2.1. Cyclic voltammetry
1.1.2.1.1. Reversibility
1.1.2.1.2. Irreversibility
1.1.2.1.3. Quasi-reversibility
2.1.2.2. Linear sweep voltammetry
2.1.2.3. Square wave voltammetry
2.1.2.4. Chronoamperometry
2.1.2.5. Chronocoulometry
2.1.3. Electrocatalysis
2.1.4. Electrochemical Impedance Spectroscopy
2.1.4.1. Data Presentation
2.1.4.2. Data Interpretation
2.1.4.3. Validation of Measurement
2.2 Chemically Modified Electrodes
2.2.1. General Methods of Modifying Electrode Surfaces
2.2.1.1. Chemisorption
2.2.1.2. Covalent Bonding
2.2.1.3. Composite
2.2.1.4. Coating by thin films
2.3 Carbon Electrodes
2.3.1. Diamond Electrode
2.3.2. Graphite Electrode
2.4 Carbon Nanotubes: General Introduction
2.4.1. Historical Perspective
2.4.2. Structure of Carbon Nanotubes
2.4.3. Synthesis of Carbon Nanotubes
2.4.4. General Applications of Carbon Nanotubes
2.5 Metallophthalocyanines: General Introduction
2.5.1. Historical Perspective
2.5.2. Structure of Metallophthalocyanines
2.5.3. Synthesis of Metallophthalocyanines
2.5.4. Electronic Absorption Spectra of Metallophthalocyanines
2.5.5. Electrochemical Properties of Metallophthalocyanines
2.5.5.1. Ring Process
2.5.5.2. Metal Process
2.5.6. General Applications of Metallophthalocyanines
2.6 Overview of Target Analytes
2.6.1. Oxygen Reduction Reaction (ORR)
2.6.2. Formic Acid Oxidation
2.6.3. Thiocyanate Oxidation
2.6.4. Nitrite Oxidation
2.7 Microscopic and Spectroscopic Techniques
2.7.1. Atomic Force Microscopy (AFM)
2.7.2. Scanning Electron Microscopy (SEM)
2.7.3. Transmission Electron Microscopy (TEM)
2.7.4. Energy Dispersive X- Ray Spectroscopy (EDS)
2.7.5. X-Ray Diffraction (XRD)
References
CHAPTER 3 Experimental
3.1 Introduction
3.1.1. Reagents and Materials
3.1.2. Synthesis
3.1.2.1. Synthesis of Nanostructured Iron(II) and Cobalt(II) Phthalocyanines
3.1.2.2. Synthesis of Iron(II) and Cobalt(II) Octabutylsulphonylphthalocyanines
3.1.2.3. Synthesis of Iron(II) tetrakis(diaquaplatinum) octacarboxyphthalocyanine (C40H24N8FeO24Pt4)
3.1.3. Purification of Multi-Walled Carbon Nanotubes
3.1.4. Electrode Modification Procedure
3.1.5. Electrochemical Procedure and Instrumentation
References
SECTION B
RESULTS AND DISCUSSION
CHAPTER 4 Microscopic, Spectroscopic, Electrochemical and Electrocatalytic Properties of Nanostructured Iron(II) and Cobalt(II) Phthalocyanine
4.1 Microscopic and Spectroscopic Characterisation
4.1.1. Comparative SEM, AFM and EDX
4.2 Electrochemical Characterisation
4.2.1. Electron transfer behaviour: cyclic voltammetry
4.2.2. Diffusion Domain Approximation Theory
4.2.3. Electron transport behaviour: Impedimetric characterization
4.3 Electrocatalytic Properties
4.3.1. Electrocatalytic reduction of oxygen
4.3.2. Electrocatalytic oxidation of thiocyanate
4.3.3. Electrocatalytic oxidation of nitrite
References
CHAPTER 5 Microscopic, Spectroscopic, Electrochemical and Electrocatalytic Properties of Iron(II) and Cobalt(II) Octabutylsulphonylphthalocyanine
5.1 Microscopic and Spectroscopic Characterisation
5.1.1. SEM characterisation
5.1.2. Characterization using UV-visible Spectrophotometer
5.2 Electrochemical Characterisation
5.2.1. Solution Electrochemistry
5.2.2. Electron transfer behaviour: cyclic voltammetry
5.2.3. Electron transfer behaviour: impedimetric characterisation
5.3 Electrocatalytic Properties
5.3.1. Electrocatalytic reduction of oxygen
5.3.1.1. Effect of scan rates
5.3.1.2. Chronocoulometric studies
5.3.1.3. Hydrodynamic voltammetry investigation
5.3.2. Electrocatalytic oxidation of thiocyanate
5.3.3. Electrocatalytic oxidation of nitrite
References
CHAPTER 6 Microscopic, Spectroscopic, Electrochemical and Electrocatalytic Properties of Iron(II) tetrakis(diaquaplatinum)octa-carboxy phthalocyanine
6.1. Microscopic and Spectroscopic Characterisation Microscopic and spectroscopic information of iron
6.1.1. UV-visible characterisation
6.1.2. Elemental analysis and mass spectroscopy characterisation
6.1.3. XRD and EDX Characterisation
6.1.4. Comparative SEM and TEM characterisation
6.2. Electrochemical Properties
6.2.1. Solution electrochemistry
6.2.2. Electron transfer behaviour: cyclic voltammetry
6.2.3. Electron transfer behaviour: Impedimetric characterisation
6.3 Electrocatalytic Properties
6.3.1. Electrocatalytic reduction of oxygen
6.3.2. Electrocatalytic oxidation of formic acid
6.3.2.1. Comparative cyclic voltammetric response
6.3.2.2. Chronoamperometry experiment
6.3.2.3. Concentration studies: Tafel analysis
6.3.2.4. Electrochemical impedance spectroscopy experiments
6.3.2.5. Tolerance to carbon monoxide poisoning
References
Conclusions and Future Perspectives
CONCLUSIONS
FUTURE PERSPECTIVES
APPENDIX A
List of scientific publications
APPENDIX B
List of scientific conferences