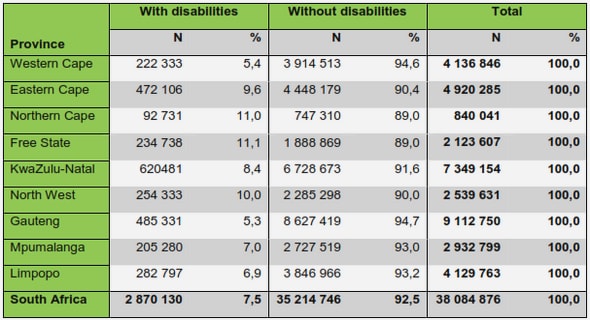
Get Complete Project Material File(s) Now! »
Hydrogen bond-mediated self-assembly
Over the last century the hydrogen bond has been the subject of extensive research and debate in various scientific branches including mineralogy, material science, inorganic and organic chemistry, supramolecular chemistry, biochemistry, molecular medicine and pharmacy [14]. The interest in this particular interaction is stimulated by its distinctive features, i.e. high selectivity and directionality. This type of bond offers the inherent ability to form supramolecular nanostructures from both inorganic and organic molecu-les.
The intermolecular hydrogen bond is the attractive interaction between the hydrogen attached to an electronegative atom of one molecule and an electronegative atom of another molecule. Consequently, a hydrogen atom is shared by two other atoms [24]. The atom to which the hydrogen is covalently bonded is called the hydrogen donor (X), whereas the other atom is the hydrogen acceptor (A). These electronegative atoms are usually oxygen, nitrogen, or fluorine, which have a partial negative charge. The hydrogen then has a partial positive charge. As a result, the acceptor with a partial negative charge attracts the hydrogen atom. The length of hydrogen bond is defined be intermediate between covalent (0.96 Å) and van der Waals (3-4 Å) bonds. It implies that the hydrogen bonds are stronger than van der Waals interactions, but weaker than covalent bonds. This bond strength depends on temperature, pressure, bond angle and environment. The strongest hydrogen interaction is anticipated when donor-hydrogen and acceptor atoms are collinear (Fig. 1.2). In a pioneering work, Whitesides et al. [9] demonstrated the principles for self-assembly of a bimolecular hydrogen-bonded network formed by Melamine (M) and Cyanuric Acid (CA) in aqueous solution. The M-CA system (Fig. 1.3a) is a proto-typical model system for molecular self-assembly in which the two compounds are complementary and interact through comparatively strong triple hydrogen bonds, i.e. 2 NH···O and 1 NH···N. This interaction results in the formation of a characteristic hexagonal “rosette” motif which consists of 3 M and 3 CA molecules shown in Fig. 1.3b. Growth of extended molecular networks of the M-CA rosette structure on Au(111) was recently characterized by high-resolution STM [25, 26] as exemplified in Fig. 1.3c. The same complementary hydrogen-bonding interactions are important in a number of other molecular systems guiding the formation of porous networks [27] and molecular chains [28] at surfaces.
This type of triple H-bonding will be the main driving force for molecular self-assemblies described in two other chapters of this thesis devoted to (i) the investigation of M-CA ordered structures on ultra thin insulating NaCl films grown on a Au(111) sur-face (Chapter 5) and to (ii) the self-assembly of Lander molecules on Au(111) which are studied for their possible ability to serve as moulds for growing metal wires (Chapter 6).
Stereochemistry in adsorption systems
One of the most important properties governing the biological and chemical interac-tions between molecules is molecular recognition. It depends not only on the ability of atoms within the interacting molecules to create specific bonding schemes like hydrogen bonds or π −π interactions but also on the positioning of their atoms in space which depentacene and SubPC. (c) Prochiral molecules: 1-nitronaphthalene and naphtho[2,3-a] pyrene.
termines their particular geometrical conformation [21]. The study of the relative spatial arrangement in molecules is a subdiscipline of chemistry called stereochemistry [29]. To this area of research belongs the very important class of chiral molecules that are non-superimposable on their mirror image by rotation and translation as shown in Fig. 1.4a. The most common type of chiral molecules possesses point chirality at a tetrahedral single atom, usually carbon, attached to four different groups. This central atom is the stereogenic (assymetric) centre of the molecule. These molecules can exist in two non-superimposable mirror forms, called enantiomers. An equal mixture of enantiomers is said to be racemic. Molecules which contain an inversion symmetry or mirror plane are achiral (Fig. 1.4b). However, some molecules may bear several chiral centers without being chiral overall. Molecular chirality was first discovered in 1848 when Louis Pasteur accidentally found out that a crystalline deposit of sodium ammonium tartrate presented equal amounts of left- and right-handed crystals [30] thus forming a racemic mixture of tartaric acid.
Over the past years a large number of experimental works were particularly devoted to surface chirality [15, 19]. The increasing interest to this field has been stimulated by the growing importance of enantioselective heterogeneous catalysis in chemical and pharmaceutical industries [31, 32]. However, surface chirality is not limited to biochem-istry. Additionally, and in many instances, thin film chirality is of primary importance for physical applications. Chiral films can possess extraordinary nonlinear optical activ-ity [33] or generate, for example, asymmetric scattering of polarized electrons [34]. As illustrated in Fig. 1.5a, chirality at surfaces can be induced by adsorption of chiral mo-lecules [35, 36]. In some cases, surface chirality can stem even from achiral molecules through their chiral ordering on the substrate (Fig. 1.5c). For instance, a pinwheel struc-ture was observed for intermixed C60 and chloro[subphthalocyaninato]boron (SubPC) [22].
Formation of amyloid fibrils by peptides
Under specific conditions, proteins and peptides can form toxic oligomers and fibril-lar aggregates accumulated in the human tissue in the shape of amyloid plaques which are associated with numerous neurodegenerative pathologies like Alzheimer’s, Hunt-ington’s, and Parkinson’s diseases [41]. The most attention-compelling finding is that inherently different peptides actually generate amyloid fibrils of similar structure.
Based on experimental results a tentative model for hierarchical organization of pep-tides into such fibers was developed [41]. As illustrated in Fig. 1.7, globular proteins undergo initially the misfolding process. In turn, unfolded proteins degrade into smaller fragments followed by their self-assembly. In a first organizational step, these fragments polymerize into long cross-β -sheets running along the fibril axis. These protofibrils are composed of parallel or antiparallel β -strands that are assembled side-by-side perpen-dicular to the fibril axis. Cross-β means that the strands of a β -sheet run perpendicular to the fibril axis. A preferential 1D growth of β -sheets can ensue from the formation of strong H-bonds parallel to their axes. Finally, these extended sheets can self-assemble
into mature fibrils stabilized by hydrophobic clustering of the side chains perpendicular to the fibril axes. Initially, the ability to form amyloid structure was assigned to the rel-atively small number of large proteins and peptides. However, it was shown that even short fragments of amyloidogenic proteins can give rise to fibrilar structure.
Adsorption and organization of biomolecules on surfaces
Over the last decade, a lot of studies were devoted to the self-assembly of biomolecules at solution-surface interfaces. It was demonstrated that templates can induce large-scale order in peptide assembly [42–44]. Such ordering might be quite different from that observed in the physiological environment. This comes from the fact that molecules adsorbed on a surface have less degree of freedom than in vitro.
An understanding of the processes driving self-assembly of biomolecules motivates the investigation of the H-bonding interaction which is known for example to determine the conformation and biochemical activity of nucleic acids. RNA and DNA strands are indeed stabilized via H-bonds provided by the complementary nucleobase pairs.
Therefore, the study of H-bonding interaction between nucleobases in the absence of any other perturbing interactions is an essential starting topic for more complex ex-plorations inolving larger molecules. This can be achieved by depositing these small biomolecules onto ultra-clean inert surfaces such as Au(111) in controlled UHV condi-tions. Such studies are relevant not only to understand the topology of large biomolecu-lar structures, but also in an attempt to comprehend the self-organization of spontaneous formed biomolecules into early precursors of life which might have occurred via ad-sorption onto natural surfaces. The problem of finding what kind of interactions occur between biomolecules adsorbed at surfaces is also crucial in the study of protein aggre-gation and amyloid fibril formation which are responsible for a number of neurological pathologies. Previous works have shown that small organic molecules such as nucleobases and amino acids bearing several kinds of interacting functional groups can form many self-assembled structures on surfaces [45–49]. For example guanine self-assembles into a H-bonded network having the same square arrangement as that found in DNA sequences which are rich in guanine [11, 50]. Thymine has been reported to form extended ordered 2D patterns and isolated chains when deposited onto the Au(111) surface [51]. Surface-induced chirality in self-assembled molecular chains of adenine has been evidenced on the Cu(110) substrate [52]. Additionally, a few studies concern the self-assembly of dipeptides onto surfaces [53–55].
STM image simulation with EHMO-ESQC method
The group of CEMES in Toulouse has developed a specific STM image calculation technique based on the EHMO-ESQC method. It is a combination of the extended Hückel molecular orbital (EHMO) theory and of the elastic scattering approach in quan-tum chemistry (ESQC). The ESQC technique uses a full molecular orbital description of STM tip-adsorbate-surface tunnel junction to calculate the corresponding current in a multichannel scattering approach between the bulk of the tip and the metallic bulk under-neath the surface [75]. It allows computing the molecular orbitals and the corresponding energy levels of the molecule adsorbed at the surface. This calculation technique has proved to be very reliable to simulate experimental STM images of various small and large molecules adsorbed on metallic and semiconducting substrates and on thin insu-lating films. Efforts are made to reduce considerably the computation time since this method requires the optimization of the atomic and orbital composition of the tip apex and of the conformation of the molecule. The calculated STM image should match as precisely as possible the corresponding experimental one in the x, y plane. There is no way to deal rapidly with an automated procedure taking into account the distance z at which the molecule is adsorbed at the surface. Therefore, automatic investigation of the molecular conformational states on surfaces is practically very slow and time consuming, and the optimization relies mainly on faster successive “manual” and not fully automated steps to reach the observed conformation.
Table of contents :
vii List of publications
ix List of abbreviations
1 Introduction
1.1 Motivation
1.2 Basic processes at surfaces
1.2.1 Molecular self-assembly
1.2.2 Hydrogen bond-mediated self-assembly
1.2.3 Stereochemistry in adsorption systems
1.3 Self-assembly of biomolecules
1.3.1 b-(Pleated) sheet
1.3.2 Formation of amyloid fibrils by peptides
1.3.3 Adsorption and organization of biomolecules on surfaces
1.4 Outline of the thesis
2 Methods
2.1 Scanning tunneling microscopy
2.1.1 The principle of STM
2.1.2 Theory of STM
2.1.3 The Aarhus STM
2.2 UHV chamber
2.3 STM image simulation with EHMO-ESQC method
2.4 Restrained molecular dynamic force field method
3 Towards Induction of Chirality in Surface Assemblies
3.1 Introduction
3.1.1 Amplification of surface chirality
3.1.2 Au(111)- 22 p 3 .
3.1.3 The molecular systems
3.2 STM of sec-butyl molecules
3.2.1 Brick-wall structure
3.2.2 Windmill structure
3.2.3 Statistical analysis
3.2.4 Bulky group conformations
3.2.5 Other windmill structures
3.3 STM study of co-deposited sec-butyl and tert-butyl molecules
3.3.1 “Nucleation” approach
3.3.2 “Intermixing” approach
3.4 Conclusion
4 Adsorption structures formed by tetrapeptides on Au(111)
4.1 Introduction
4.1.1 Amyloid fibril formation by KFFE tetrapeptides
4.1.2 Previous UHV STM studies of amino acids and peptides
4.1.3 The molecular systems
4.2 Parallel row structure formed by KFFE and KVVE
4.2.1 STM results
4.2.2 Restrained MDFF structure-calculation
4.3 Antiparallel row structure formed by KVVE
4.3.1 STM results
4.3.2 Restrained MDFF structure-calculation
4.4 Other structures formed by KVVE or KFFE
4.4.1 Zig-zag like structure of KVVE
4.4.2 Cross-linked row structure of KVVE
4.4.3 Windmill structure of KFFE
4.5 Conclusion
5 Molecular Self-Assembly on a Thin Insulating Film
5.1 Introduction
5.2 Experimental procedure
5.3 Growth of NaCl on Au(111) substrate
5.4 Self-assembly of M and CA on NaCl/Au(111)
5.5 Conclusion
6 Hydrogen bond induced structures of Lander molecules
6.1 Introduction
6.2 Molecular systems
6.2.1 Lander-DCI molecules
6.2.2 Lander-DAT and ND molecules
6.3 Experimental procedure
6.4 Self-assembly of Lander-DCI
6.5 Self-assembly of Landers DAT and ND
6.5.1 Anticipated interaction of Lander-DAT
6.5.2 1D-chain structure of Lander-DAT
6.5.3 H-bonded 2D structures of Lander-DAT
6.5.4 Comparison with 2D structure of Lander-ND
6.6 Co-adsorption of Lander-DCI and DAT
6.6.1 Short molecular chains at step edges
6.6.2 2D supramolecular network
6.7 Conclusion
7 Summary and outlook
Bibliography