Get Complete Project Material File(s) Now! »
Chapter 2 ZnO as a semiconductor
Introduction
ZnO is a wurtzitic compound semiconductor with a wide and direct bandgap of 3.4 eV at room temperature. This material occurs naturally as n-type with the native defects particularly zinc interstitials, and oxygen vacancies being proposed to be the source of n-type conductivity [1]. Some reports claim that the n-type conductivity is due to some impurities introduced during material growth predominantly hydrogen and other group I related elements, arguing that the oxygen vacancy is too deep to contribute a significant concentration to the n-type conductivity of ZnO [3,4,5,6,7]. The effects of hydrogen in ZnO have been studied. It has been proposed that hydrogen in ZnO is a shallow donor [2,8,9] and can form several bond structures which include the BCII and BC┴ configurations [2]. However the realization of p-type ZnO still remains a challenge as it lacks reproducibility [10, 11].
Crystal structure
ZnO is a highly ionic compound semiconductor which crystallizes into three common lattice structures, and these are; the rock salt, zinc blende and the wurtzite structure. The wurtzite structure is the most thermodynamically stable one at ambient conditions [12] i.e. for bulk ZnO crystals. This structure can be considered as two interpenetrating hexagonal-close-packed lattices, characterized by two interconnecting sublattices of Zn2+ and O2- such that each Zn ion is surrounded by a tetrahedral of O ions and vice versa [13]. The tetrahedral arrangement gives rise to the polar symmetry along the hexagonal axis. Four common terminations for the wurtzitic ZnO structure exist and these are; Zn-polar terminated (0001) and the O-polar terminated ( 000 1 ) faces (c-axis oriented), the non-polar (1120 ) (a-axis) and (10 10 ) faces which both contain an equal number of Zn and O atoms. Due to the difference in face terminations, the polar faces have different physical and chemical structures; with the O-polar face having a unique electronic structure from the other three [13]. The wurtzite structure of ZnO is shown in Figure 2.1.
Material growth techniques
ZnO is available as bulk single crystals and also as thin films. Bulk material is grown using the hydrothermal, chemical vapour transport (CVT), and melt growth techniques, while thin film material is grown using the pulsed laser deposition, metalorganic chemical vapour deposition (MOCVD), metalorganic vapour phase epitaxy (MOVPE), just to mention a few. In this particular study, main focus is on melt grown bulk ZnO single crystals. Traditional melt growth processes rely on material growth from a melt or liquid. They are associated with three main hindrances, which include melt containment, ZnO decomposition at atmospheric pressure near the melting point, and crystal contamination [21]. These problems are however eliminated in the melt growth technique used by Cermet [21]. The melt growth technique produces high quality material that is free from solvent related impurities and thus minimal defects. The other advantage of this technique is the high material growth rate [21].
Properties and Applications
Due to its wide bandgap, this material finds its use in several applications some of which include the fabrication of ultraviolet detectors and daylight-blind UV detectors, transparent conducting oxides, acoustic wave devices, light emitting diodes, laser diodes, high frequency electronic devices, varistors, piezoelectric transducers, and paints. ZnO has an excitonic binding energy of 60 meV at room temperature and is compatible with the conventional chemistry etching similar to that used in Si technology which gives it more advantages over other wide band gap materials, e.g. GaN.
The use of ZnO in the electronic and optoelectronic industries requires the fabrication of highly stable good quality metal/ZnO contacts. Fabrication of good quality rectifying contacts has been a problem since the early discoveries of the material and still remains a challenge as different Schottky barrier heights have been reported for the same metal deposited on ZnO. Reports have indicated the dependence of contact quality on cleaning procedures used other than on the metal work function [22]. Polarity of the material [23] (in the case of bulk material) and the deposition techniques used [24] have also been reported to affect contact quality. This is valid since different cleaning procedures will influence the surface states, hydrocarbon and hydroxide surface contamination, oxygen chemisorption, subsurface defects and surface morphology due to the chemical reactions taking place at the surface. The above-mentioned will in turn affect the metal-surface reactions and hence the contact quality.
Table 2.2 shows the variation of measured current voltage (IV) parameters with cleaning procedures and deposition techniques from selected references. These parameters have been obtained at room temperature.
A dramatic improvement of contact quality has been reported after the treatment of the material with hydrogen peroxide [26,27,32,33,34,35]. The study of the effects of deposition techniques has not been extensively carried out. High quality metal/ZnO contacts are of importance in the fabrication of UV detectors and also in the characterization of deep level defects in semiconductors.
Modifications of the electrical properties of ZnO after annealing in different ambient conditions have been studied using Hall Effect measurements [36,37,38], photoluminescence and DLTS measurements [40,41,42]. The Hall Effect measurements have indicated an increase in surface conductivity after hydrogen annealing [43, 44] which influences device characteristics. A reduction of the shallow donor concentration in ZnO after Ar annealing has been reported [45]. Passivation of deep level defects in ZnO after hydrogen annealing has also been observed using the PL measurements [44]. Oxygen annealing at high temperatures is also of very high significance as it helps in eliminating surface adsorbents. At the same time, it leads to surface thermal decomposition which is an ideal method for preparing the material for surface electronic studies. This clearly indicates that the electronic and optical properties of ZnO can be easily altered by thermal treatments to suit the needs of the researcher. The fact that ZnO allows for band gap engineering by alloying it with MgO and CdO to increase and decrease its bandgap energy, respectively [46] is an important aspect since it allows for tuning of the material’s optoelectronic properties for use in selected wavelengths of the electromagnetic spectrum for selective light sensing purposes.
Metal/ZnO contacts
Introduction
Metal/semiconductor contacts are important to the microelectronics industry as well as in the characterization of fundamental properties of semiconductors using junction techniques e.g. DLTS. The quality of a metal/semiconductor contact determines its performance as poor contacts result in undesired effects e.g. high leakage currents and low barrier heights. Metal/semiconductor quality is determined by a number of factors, some of which include the mismatch between the Fermi-energies of the metal and semiconductor, the semiconductor processing steps (cleaning and annealing) prior to contact fabrication and the contact fabrication techniques. These contacts are considered to be ohmic or rectifying (Schottky) as governed by their response to an applied external bias.
Ohmic contacts
A contact is said to be ohmic if the barrier formed from the intimate contact of a metal and semiconductor is zero. In an ideal ohmic contact, charge carriers are free to move in either direction across the contact with minimal or no resistance. To fabricate such a contact on an n-type semiconductor, we require that the metal work function must be closer to or smaller than the semiconductor electron affinity. Formation of such a contact is illustrated in Figure 2.2.
Bringing the metal into intimate contact with the semiconductor will result in Fermi-level alignment as shown in Figure 2.2b. This raises the semiconductor electron energies relative to the metal, reducing the barrier to electron flow between the metal and semiconductor. However under real situations, ohmic contact formation is affected by external factors, some of which include; conditions under which the contacts are fabricated, surface states, interface states, and also reactions between the metal and semiconductor. Ohmic contacts are classified into tunnel, and annealed and alloyed contacts; with the tunnel contacts being the most practical ones. Tunnel contacts possess a positive barrier at the metal/semiconductor interface, but also have enough doping in the semiconductor such that there is only a thin barrier separating the metal from the semiconductor. This thin barrier allows carriers to easily tunnel through the interface. For an ohmic contact, under any applied biasing conditions, the IV characteristics must always obey Ohm’s law, since there is a very thin barrier for electron flow in any given direction, i.e. metal to semiconductor or semiconductor to metal.
Schottky contacts
If a metal-semiconductor contact can rectify signals in the forward biased mode and allows negligible or no current to flow in the reverse bias, it is referred to as a Schottky contact. Ideal Schottky contacts are formed when a difference in potential exists between the Fermi energy of the metal and the band edge where majority charge carriers reside (ignoring the effects of surface and interface states). The formation of a Schottky contact is illustrated in Figure 2.3. The difference in potential between the Fermi-energy of the metal and semiconductor gives the barrier height, . For an n-type semiconductor, the barrier height is given theoretically as [47];
where is the metal work function, is the electron affinity of the semiconductor. In real Schottky contacts, the barrier height is affected by interfacial layers, surface states, defects and chemical reactions which also determine the quality of the contacts.
Current-voltage (IV) measurements
The current voltage technique offers a good method for evaluating the quality of a Schottky contact by determining the reverse and forward current that flows through the device under different biasing conditions. This enables us to deduce the current transport mechanisms that dominate the conduction process within the device. Figures of merit which include barrier height, ideality factor and series resistance can also be obtained.
In this section, a couple of IV characteristics obtained from metal/ZnO structures are presented and discussed. This section aims to point out some of the factors affecting the quality of the contacts, in as much as the fabrication techniques and material processing steps are concerned.
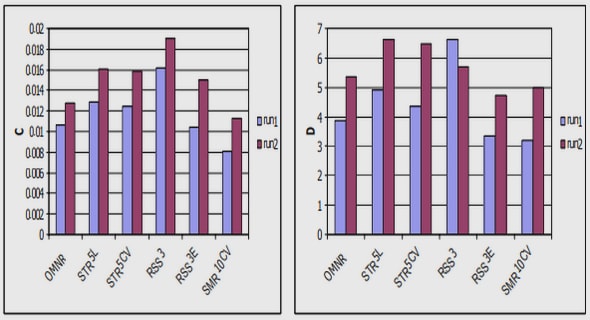
A typical current-voltage characteristic of metal/ZnO Schottky contacts is illustrated in Figure 2.4. These contacts show a strong dependence on the contact fabrication technique used since the material processing steps are the same. The straight line shows the linear fit to the data by assuming a pure thermionic emission model. The current flowing through the metal semiconductor contact is given by [48],
Deviations from linearity at high applied voltage are due to the effects of series resistance.The resistive evaporation technique proves to produce better quality contacts with very low leakage currents and a higher degree of rectification compared to the e-beam deposited contacts. Table 2.3 shows some of the parameters extracted from the resistively deposited and e-beam deposited contacts by fitting the data to a pure thermionic emission model.
Chapter 1 Introduction
References
Chapter 2 ZnO as a semiconductor
2.1 Introduction
2.2 Crystal structure
2.3 Material growth techniques
2.4 Properties and Applications
2.5 Metal/ZnO contacts
2.5.1 Introduction
2.5.2 Ohmic contacts
2.5.3 Schottky contacts
2.5.3.1 Current-voltage (IV) measurements
2.5.3.2 Capacitance-voltage (CV) measurements
2.5.4 The Depletion layer
Summary
References
Chapter 3 Defects in semiconductors
3.1 Introduction
3.2 Point defects
3.2.1 The Oxygen vacancy
3.2.2 The Zinc vacancy
3.2.3 The Zinc interstitial
3.2.4 Negative-U and Metastability
3.3 Extended defect
3.3.1 Line defects
References
Chapter 4 Defect characterization
4.1 Introduction
4.2 Emission and capture of carriers from deep levels
4.3 Deep level transient spectroscopy, DLTS
4.4 Defect Depth profiling
4.5 Laplace DLTS
Summary
References
Chapter 5 Experimental Techniques
Introduction
5.1 Sample cleaning
5.2 Contact fabrication
5.3 Current-Voltage and Capacitance-Voltage measurements
5.4 Deep Level Transient Spectroscopy (DLTS) and Laplace-DLTS
5.5 Sample annealing
5.6 Proton irradiation
References
Chapter 6 Results and discussions
6.1 Defects in ZnO
6.2 Electron-beam induced defects
6.3 Annealing studies of ZnO using Hall Effect, IV and DLTS
6.4 Irradiation induced defects
Chapter 7 Conclusions
Chapter 8 Future Work
GET THE COMPLETE PROJECT