Get Complete Project Material File(s) Now! »
Definition of HSCL-25
The HSCL-25 is a self-report questionnaire on the existence and severity of both anxiety and depression symptoms during the previous week, used to identify psychiatric illness in primary care (49). It includes 25 items: 10 items about anxiety and 15 about depression (50). The patient is considered as a “probable psychiatric case” if the mean rating on the HSCL-25 is ≥ 1,55. A cut-off value of ≥ 1,75 is generally used for diagnosis of major depression defined as “a case, in need of treatment” (51,52). The HSCL-25 was used in primary care in family planning services, among refuges and among migrants (53–55).
For the translation to retain the same meaning as the original, a Forward-Backward translation (47)(56) was conducted following a formal consensus method, the Delphi procedure. Formal consensus is an appropriate method to reach a solid consensus transparently on a little investigated subject (57). Delphi procedure, reliable and efficient is used frequently in health care as a rigorous way to reach consensus in defined clinical areas (58-61). It is a systematic interactive method which involves a panel of experts using iterative procedures. It can be done quickly to make a single convergent final recommendation. This process requires to follow four rules: anonymity of participants (ensures responses reliability and avoids contamination), iteration (allows participants to refine their views in the light of the progress of the group’s work), control feedback (under the responsibility of the investigator), statistical aggregation of group’s responses to allow a quantitative and qualitative analysis of the data (46)(62-64).
Consents and anonymity
In each country, the Investigator was the National investigator (NI). He asked the participants for their signed consent (annex 2), anonymized the expert responses and delivered an identification number for later identification. The name of each expert was not transmitted to others (46). Only NI’s consent was sent to the team of investigators (annex 3, annex 4). As the study involved no patient, it didn’t require an ethics committee‘s decision.
Participants
Pilot Team (PT): The EGPRN French team was familiar with the Delphi methodology. It requested the consent of each national investigator as well as voluntary participation in the study and an absence of conflict of interest statement. It ensured that the whole process followed the protocol. It didn’t take part in the translation phases or in Delphi rounds. The Forward-Backward translation had to be validated by the daily board of the study, composed of members of EGPRN all active within the research process.
National Investigators (NI): The NIs’ were in charge of recruiting translators and experts. They acted between each phase and between two Delphi rounds. They didn’t act when a Delphi round was running. Translators: The NIs’ selected translators to make up two translation teams. Translators had to be knowledgeable about health care terminology. The Forward translation team involved one member of the GPs’ research group and one official translator in every country. The Backward translation team involved one (or two) GP(s’) and one official translator (46). The two teams should not have involved the same person (65).
Experts panel: Initially, 20 to 30 experts were recruited in order to keep at least 15 participants until the end of the last round (according Delphi’s quality standards criterias. The selection criteria for every expert were: being native to country of origin and speaking their native language; being an English speaker; being a GP. Over half had to have teaching or research activities. In order to assess the representativeness of the panel by its maximum variation, the experts informed their gender, area of practice, years of practice and publications.
Forward Translation
The PT sent the HSCL-25 original English version to the NI who sent it to the Forward translation team. This team translated HSCL-25 from English to language of the country aiming to retain the same meaning as the original. There were 32 elements to translate: the introductory sentence, the 25 items, the 4 adjectives corresponding to the proposals, and two sentences relating to the use of the survey (explanatory sentence). An exception for Germany, a German version of the HSCL-25 already existed. The forward translation was not performed. The Delphi procedure was applied directly to the German version. This made it possible to verify the quality of this translation for a qualitatively representative panel of GP.
Delphi rounds
At the beginning of the first round, NIs’ sent by mail the original English version and translated version in Galician, Castilian, Catalan, Bulgarian, Greek, Croatian, Italian, French, German, Polish with the entire rules of procedure. GPs’ experts received records individually. NIs didn’t use a mailing list in order to assure anonymity which increases the reliability of responses and avoids contamination (discussion between experts).
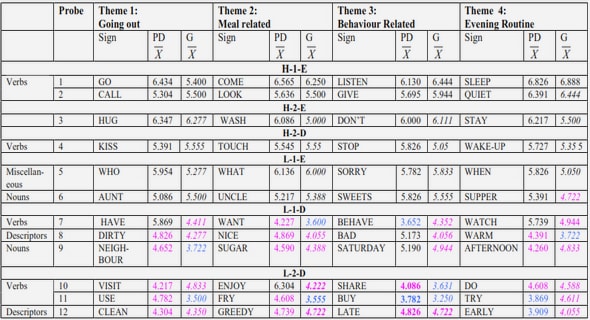
Experts expressed their level of agreement on each proposal by using a 1 to 9 Likert scale. This Likert scale was an agree/disagree scale, symmetric, odd, that measured the intensity of their feelings on each proposal, taking into account the maintenance of the meaning between the original and the translation proposal, the ergonomics and the ease of understanding. Experts rated the proposal from 1 (absolutely no agreement) to 9 (fully agreement) and had to comment when rating less than 7. Consensus was defined for an excerpt’s translation when it was rated 7 or above by over 70% of the panel (66), so it was accepted directly and didn’t enter the following rounds; if not (proposal didn‘t reach consensus), the NI and the Forward official translator synthesized experts comments to propose a new translation proposal for this excerpt. Time between two rounds had to be less than four weeks. The following round began when the NI sent to the experts separately for each excerpt that didn’t reach consensus: the original English version, the unaccepted proposal with all the experts’ comments on this proposal, the new proposal. Experts rated the new proposal in the same way as the first round. The following rounds rolled out in an identical manner. This process was repeated until all excerpts found a consensual translation. The number of rounds was not limited (67). At the Delphi procedures end, there were a HSCL-25 consensual versions in different European languages.
Backward translation
NI sent the final Galician, Castilian, Catalan, Bulgarian, Greek, Croatian, Italian, French, German or polish version of HSCL-25 to the backward translation team who had to translate it into English. The translators should not have the knowledge of the original version (blind-back translation principle). Finally, he sent the Backward English version to the PT (68).
Table of contents :
Résumé
Abstract
1-Introduction
2- Materials and methods
2.1. Definition of HSCL-25
2.2. Consents and anonymity
2.3. Participants
2.4. Forward translation
2.5. Delphi rounds
2.6. Backward Translation
3- Résults
3.1. Forward
3.2. Panel
Table 1: leader panel
Table 2: Experts’ panel
3.3. Number of delphi round
Table 3: Results of the first Delphi round
3.4. For each language
3.4.1. Galician
3.4.2. Castille
3.4.3. Catalan
Table 4: Forward/backward: Galicia, Catalan, Castilian
3.4.4. French
3.4.5. Italian
3.4.6. Bulgarian
Table 5: Forward/backward: Italian, French, Bulgarian
3.4.7. Croatian
3.4.8. Greek
3.4.9. Polish
Table 6: Forward/backward: Croatian, Greek, Polish
3.4.10. German
4. Discussion
4.1. Main results
4.2. Teaching, research and practice future applications
4.3. Strenght and limits
4.3.1. Strenght
4.3.2. Selection bias
4.3.3. Information bias
5- Conclusion
6- Bibliography