Get Complete Project Material File(s) Now! »
CHAPTER 2: GENOTOXICITY TESTING
BACKGROUND
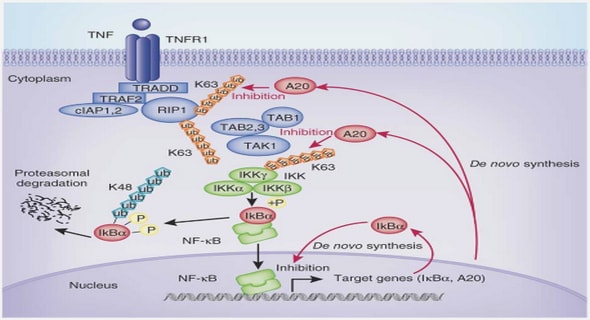
In a study by You-Cheng et al., (2008), the genotoxic potential of humic acid (HA) is assessed using human peripheral blood lymphocytes, using comet and sister chromatid exchange assays respectively. Their study suggested that HA can induce genotoxicity in human lymphocytes as evidenced by DNA damage and that HA induced changes in Ca2+- homeostasis and ROS/RNS production appear to be the main pathways of this genotoxicity induction. This demonstration of HA genotoxicity lead to their hypothesis that HA could induce cancer by disrupting genetic integrity (You-Cheng et al., 2008). As described earlier, humic substances are divided into fulvic acids, humic acids and humin on the basis of the solubility in water as a function of pH. Fulvic acid is the fraction that is soluble in water under all pH conditions and is in general lower in molecular size and has lower colour intensity than humic acids. The average molecular weight of humic substances may range from 500 to 5000 Da for fulvic acid and 3000 to 1000 000 Da for humic acid (Sparks, 2003). The potential genotoxicity of CHD-FA was tested using the in vitro micronucleus test as part of the battery tests required to determine potential genotoxicity in combination with a bacterial reverse mutation test (Ames test) which was conducted by Dr G Jooné (data on file Department of Pharmacology, University of Pretoria, South Africa).The presence of micronuclei (MNi) in cultured human cells was reported as early as the 1960s (Elston, 1963). The in vitro micronucleus test is a robust quantitative assay of chromosome damage due to the development of the cytokinesis-block technique which has eliminated the confounding effects on MNi expression by the cytostatic effects caused by poor culture conditions, treatment effects, cell senescence and variability in mitogen response in the lymphocyte test system (Fenech and Morley, 1985, 1986).In the cytokinesis-block micronucleus (CBMN) assay, scoring of MNi allows for the differentiation of post-divided cells that are accumulated and recognised by their binucleated appearance from the mononucleated cells that did not divide during the in vitro culturing period.This technique has become an attractive tool for genotoxicity testing because of its capacity to detect not only clastogenic and aneugenic events but also some epigenetic effects and its simplicity of scoring, accuracy, wide applicability in different cell types and amenability to automation (OECD 1997).
This in vitro micronucleus assay is a mutagenicity test system that assesses chemicals for their ability to induce the formation of small membrane-bound DNA fragments such as micronuclei in the cytoplasm of interphase stage cells during cell division. These micronuclei may originate from acentric fragments (chromosome fragments lacking a centromere) or whole chromosomes that are unable to migrate to the poles with the rest of the chromosomes during the anaphase of cell division (Kirsch-Volders, 1997). The advantages of this technique are well defined:
1. It is a multi-target genotoxic endpoint, predictive for cancer (Bonassi et al., 2010)
2. It is amenable for automation (Decodier et al., 2010)
3. It allows good extrapolation for potential limits of exposure or thresholds (Elhajouji et al., 2010)
4. MNi can be scored easily, measured in a variety of systems, in vitro (Hayashi et al., 2007).
There is an extensive amount of data to support the validation of the in vitro micronucleus assay using various cell lines or human lymphocytes, which include, in particular, the international validation studies co-ordinated by the French Society of Genetic Toxicology (Bonassi et al., 2010; Decodier et al., 2010 Elhajouji et al., 2008, 2010 Hayashi et al., 2007; Dertinger et al., 2010;Fenech 2000) and the reports of the international in vitro micronucleus assay working group (Fenech and Morley, 1985, 1986). Implementation of this assay in the battery of tests, such as sister chromatid and comet assays, for the hazard and risk assessment of potential mutagens/carcinogens is therefore fully justified.
Declaration
Acknowledgments
Abstract
List of Figures
List of Tables
Abbreviations
1 CHAPTER 1: LITERATURE REVIEW
1.1 THE DRUG DEVELOPMENT PROCESS
1.2 HUMIC SUBSTANCES AND MEDICINAL APPLICATIONS
1.3 MICRONUCLEUS AND GENOTOXICITY
1.4 INFLAMMATION
1.5 ATOPIC DERMATITIS
1.6 LIQUID CHROMATOGRAPHY TANDEM MASS SPECTROMETRY AND BIOMARKER DISCOVERY
1.7 STUDY AIM
2 CHAPTER 2: GENOTOXICITY TESTING
2.1 BACKGROUND
2.2 METHODS
2.3 RESULTS
2.4 DISCUSSION
2.5 CONCLUSION
3 CHAPTER 3: CLINICAL SAFETY AND EFFICACY
3.1 INTRODUCTION AND BRIEF STUDY OUTLINE
3.2 HYPOTHESES
3.3 STUDY OBJECTIVES
3.4 STUDY PLAN AND EVALUATION
3.5 SELECTION OF VOLUNTEERS
3.6 TREATMENTS
3.7 WITHDRAWALS
3.8 ADVERSE EVENTS
3.9 STATISTICAL METHODOLOGY AND ANALYSIS
3.10 ETHICAL CONSIDERATION
3.11 MEDICAL CONTROL COUNCIL (MCC)
3.12 RESULTS
3.12.1 Acute Toxicity
3.12.2 Sub-Acute Toxicity
3.12.3 Proof of concept
3.13 DISCUSSION
3.14 CONCLUSION
4 CHAPTER 4: TOPICAL EFFICACY OF CHD-FA AS AN ANTIINFAMMATORY AGENT
4.1 INTRODUCTION AND BRIEF STUDY OUTLINE
4.2 HYPOTHESIS
4.3 STUDY OBJECTIVE
4.4 STUDY DESIGN AND EVALUATION
4.5 RESULTS
4.6 DISCUSSION
4.7 CONCLUSION
5 CHAPTER 5: LC-MS/MS INVESTIGATION OF CHD-FA BIOMARKERS IN PLASMA
5.1 INTRODUCTION
5.2 MATERIALS AND METHODS
5.3 RESULTS
5.4 DISCUSSION
5.5 CONCLUSION
6 REFERENCES
7 RESEARCH OUTPUTS
8 SUPPLEMENTARY DOCUMENTATION
9 PATIENT INFORMATION LEAFLET AND INFORMED CONSENT
PATIENT INFORMATION LEAFLET AND INFORMED CONSENT