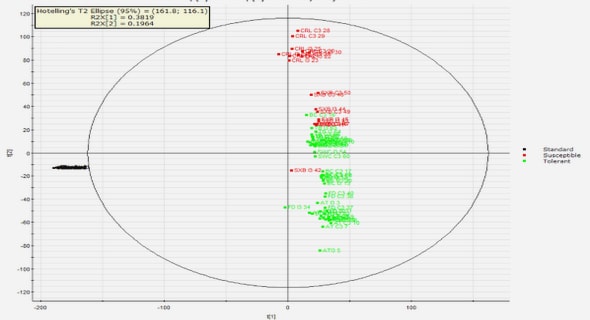
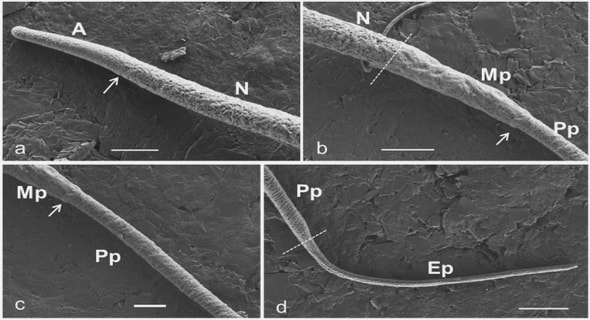
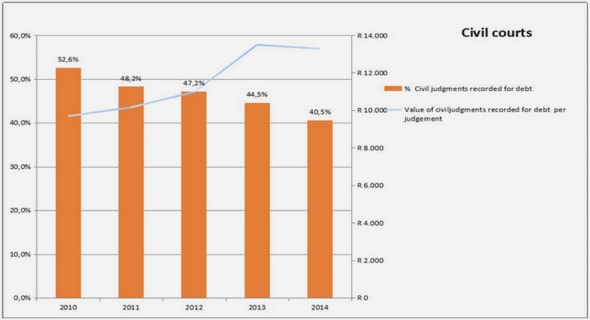
Get Complete Project Material File(s) Now! »
Horizontal gene transfer and the implications for rhizobial taxonomy
The transfer of genes between lineages, referred to as horizontal gene transfer, complicates phylogenetic reconstruction, because some species are chimeric, with different phylogenetic histories for different parts of the genome (Eisen, 2000). Genes can be transferred by conjugation, tra~sformation or transduction (Schloter et al., 2000; Dutta and Pan, 2002).
Horizontal gene transfer changes the microbial genome. Novel metabolic capabilities are conferred to the genome through horizontal gene transfer and this enable the recipient organism to explore new ecological niches (Dutta and Pan, 2002). Horizontal gene transfer could initiate diversification of bacterial lineages (Lawrence, 1999; Schloter et al., 2000, Dutta and Pan, 2002).
It is now clear that the genes for pathogenic or symbiotic interactions with eukaryotes are acquired through horizontal gene transfer (Finan, 2002). The genes can be located on plasmids or on the chromosome as genomic islands. In most cases the genomic islands loose their transmissibility (Finan, 2002), but in the case of M loti strain R7A, it was shown that this strain still had the necessary machinery to transfer the symbiosis island (Sullivan et aI., 1995; Sullivan and Ronson, 1998). Rosse1l6-Mora and Amann (2001) argued that if it is known that characters, especially extrachromosomally coded characters, which affect phenotype have been acquired through horizontal gene transfer, these characters should be excluded from taxonomic studies.
Functional or essential genes like the ribosomal genes are not usual candidates for horizontal gene transfer, since the recipient genome already has functional copies (Lawrence, 1999).
The native genes have co-evolved with the rest of the cellular machinery and it is thus unlikely to be displaced, which makes it difficult for the novel genes to persist in the recipient genome. However, horizontal gene transfer of essential genes does occur. Thermomonospora has two functional copies of rRNA genes; one of the copies was obtained by horizontal gene transfer (Lawrence, 1999). Bacillus megaterium harbours a functional rRNA operon on a small plasmid, which might be transferable to other bacteria (Broughton, 2003). In the family, Rhizobiaceae the horizontal transfer of rRNA genes and recombination among species of the same genus did occur (Eardly et al., 1996 and Parker, 2001). Gaunt et al. (2001) reconstructed phylogenetic trees from atpD (p-subunit of the membrane ARP synthase) and recA (encodes part of the DNA recombination and repair system) sequence data. The researchers found that the trees supported the 168 rDNA sequence based classification of rhizobia, but possible recombination events within the genera were found (Gaunt et al., 2001). Turner and Young (2000) found that glnI (glutamine synthetase found in all prokaryotes) phylogeny and 168 rDNA phylogeny were in broad agreement. However, incongruence between the 168 rDNA phylogeny and glnII (glutamine synthetase found in eukatyotes and some prokaryotes) phylogeny were found. The incongruence might be explained by horizontal gene transfer to Bradyrhizobium, Rhizobium galegae and Mesorhizobium huakuii. The researchers also found evidence of horizontal gene transfer within the genus Mesorhizobium (Turner and Young, 2000). A new report by Van Berkum et al. (2003) showed incongruence between phylogenetic trees
reconstructed from 168 rDNA and those reconstructed from IG8 and 238 rDNA sequence data. Analysis of the sequence data identified possible recombination events between short segments of the 168 rRNA genes of B. elkanii with Mesorhizobium species and between species of Sinorhizobium and Mesorhizobium species (Van Berkum et al., 2003). It is clear that phylogenetic trees based on 168 rDNA sequence data should be used with caution, but the trees can still give a basic idea of the taxonomic position of an isolate. Broughton (2003) suggested that authors should wait for the sequence data of other conserved genes to become available before new changes are made to the taxonomic groupings of rhizobia. This opinion was also shared by Van Berkum et al. (2003). Rhizobial taxonomists should thus strive for a taxonomy that is stable: a name should not be a source of error, a name should be maintained for as long as possible, a name should be accepted by evety rhizobia! scientist and finally the name should be unambiguous (Broughton, 2003).
The current taxonomy of the root-nodulating bacteria
Jordan (1984) included all rhizobia in the family Rhizobiaceae in the a-2-subgroup of the Proteobacteria. The rhizobial taxonomy changed significantly since 1984 with the development of new techniques and the study of more diverse legumes. On the website (http://www.cme.msu.edu/bergeys/outline.prn.pdf) of Bergey’s manual rhizobia are included in several different families. Rhizobium, Allorhizobium and Sinorhizobium are placed in the family Rhizobiaceae. Mesorhizobium is grouped in the family « Phyllobacteriaceae », however Gaunt et al. (2001) found little support for this in their study on the atpD, recA and 16S rDNA sequence data of the a-Proteobacteria. Bradyrhizobium are placed in the family « Bradyrhizobiaceae », while Azorhizobium and the genus Devosia in which a newly nodulating species have been described belong to the family Hyphomicrobiaceae. The other nodulating species of the a-Proteobacteria belong to the genus Methylobacterium in the family « Methylobacteriaceae » (Bergey’s manual taxonomic list: http://www.cme.msu. edu/bergeys/outline/prn.pdt).
Recently, it became clear that the ability to nodulate and fix nitrogen is not restricted to the a- Proteobacteria, but that several species in the ~-Proteobacteria acquired the ability as well. The two genera involved Burkholderia and Ralstonia belong to the families « Burkholderiaceae » and « Ralstoniaceae » respectively in the order « Burkholderiales » (Bergey’s manual taxonomic list: http://www.cme.msu.edu/bergeys/outline/prn.pdt). In 2001, Young et al. proposed the emended description of Rhizobium (Fig. 5.1) to include the previous genera Allorhizobium, Agrobacterium (genus of plant pathogens) and Rhizobium.
This proposal was based on high 16S rDNA sequence similarity values and no clear phenotypic differences between the three genera (Young et al., 2001). With specific reference to the inclusion of Agrobacterium in the genus Rhizobium, Broughton (2003) stated that it is important for a bacterial name to reflect the reality of its symbiotic or pathogenic nature. Even if the true nature (symbiotic or pathogenic) of the organism is not reflected in its 16S rDNA sequence (Broughton, 2003). The International Committee on Systematics of Prokaryotes (2002) stated that the latest proposal of a new name does not mean that it has preference over the older name. The committee suggested that individual experts decide which name they want to use.
CHAPTER 1 INTRODUCTION
CHAPTER 2 NODULATION: PROCESS AND APPLICATION
1. Introduction
2. Nodulation
2.1 The nodulation process
2.2 Genetics of nodulation
2.3 Factors unrelated to Nod-factor structure, which influence nodulation
3. Evolution of the ability to nodulate in legumes
3.1 Possible explanations for nodule development in legumes
3.2 Evolution of the bacterial symbiont
4. Application of the legume-rhizobium-Iegume symbiosis
4.1 Inoculation of legumes
4.2 Co-inoculation
5. Other nitrogen fixation associations
5.1 Frankia and actinorhizal symbiosis
5.2 Gunnera sp. – Nostoc symbiosis,Azo/la sp. –Anabaena symbiosis and cycads (Encephalartos spp.) – Nostoc symbiosis
5.3 Associative nitrogen fixation
CHAPTER 3 THE PLANT PARTNER: THE GENUS CYCLOPIA
1. introducing the legume family Fabaccae
2. the tribe Podalyrieae
3. The genus Cyclopia
3.1 Distribution of the genus Cyclopia
3.2 Honeybush tea
CHAPTER 4 POLYPHASIC TAXONOMY
1. what is polyphasic taxonomy
2. Genomic techniques
2.1 Determination of the DNA base ratio
2.2 DNA homology
2.3 DNA typing techniques
3. Phenotypic technique
CHAPTER 5 TAXONOMY OF THE ROOT-NODULATING BACTERIA
1. The polyphasic taxonomy of root-nodule bacteria
2. Horizontal gene transfer and the implications for rhizobial taxonomy
3. The current taxonomy of the root-nodulating bacteria
3.1. The genusAllorhizobium
3.2 The genusAzorhizobium
3.3 The genus Bradyrhizobium
3.4 The genus Rhizobium
3.5 The genusMesorhizobium
3.6 The genus Sinorhizobium
3.7 Root-nodulating species from other genera
CHAPTER 6 DETERMINATION OF THE DIVERSITYOF ROOT-NODULATING BACTERIA ASSOCIATEDWITH CYCLOPIA SPP.
CHAPTER 7 PHYLOGENETIC, SYMBIOTIC ANDPHENOTYPIC CHARACTERISATION OF SOME BURKHOLDERIA SPP. ISOLATES
CHAPTER 8 PHYLOGENETIC ANDSYMBIOTIC CHARACTERISATIONOF THE a-PROTEOBACTERIA CYCLOPIA ISOLATES
CHAPTER 9 CONCLUSIONS
CHAPTER 10 REFERENCES